FUJIFILM Wako Chemicals U.S.A. Corporation
FUJIFILM Wako Chemicals U.S.A. Corporation continues to maintain a proud history of product quality and customer service through the establishment of our LAL Division, and the introduction of our new PYROSTAR™ ES-F line for the detection of bacterial endotoxin.
FUJIFILM Wako Chemicals U.S.A. Corporation (FUJIFILM Wako) is recognized around the world as a trusted supplier of pure chemicals and reagents. FUJIFILM Wako PYROSTAR™ ES-F series offers a simple, accurate platform for the detection of bacterial endotoxin. This endotoxin-specific LAL reagent is accompanied by a suite of complementary products to offer a complete solution for your endotoxin testing needs.
FUJIFILM Wako provides a line of reagents formulated to perform each of the three endotoxin detection assays: the gel clot assay, the kinetic turbidimetric assay (KTA) and the kinetic chromogenic assay (KCA).
Download our catalog of LAL REAGENT PRODUCTS For the Detection of Bacterial Endotoxin here
Product News
View All Product News
Products
Toximaster® QC8 Software
Pharmaceutical
The Toximaster® QC8 software supports data processing based on the protocols complying with three types of pharmacopeia (USP/EP/JP) for bacterial endotoxin testing. This software is compliant with FDA 21 CFR Part 11 ERES (electronic records/electronic signature), which requires the proper information from all parties involved in the testing of the sample. Toximaster® QC8 offers excellent audit trail capabilities, can provide statistical processing of means and standard deviations, and has robust data processing functions. These data processing functions can be performed by defining sample types such as standards, controls, and test samples.Toxinometer® ET-7000 Endotoxin Measurement System
Pharmaceutical
The Toxinometer® ET-7000 is a computer-operated kinetic incubating tube reader, designed to be exceptionally user-friendly. State-of-the-art expansion modules can be connected to allow for endotoxin testing in a wide range of fields and sample quantities. The Toxinometer® ET-7000 can perform all LAL testing methodologies (gel clot, kinetic turbidimetric, and kinetic chromogenic assays). The instrument has 16 wells for single-test configuration in order to easily detect erroneous readings or contamination associated with each well. The kinetic incubating tube reader has uniformity temperature settings of 30°C and 37°CES Buffer
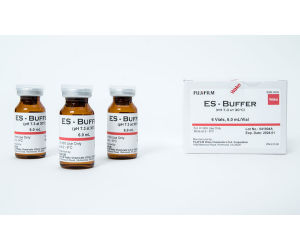
Pharmaceutical
The ES Buffer is an endotoxin-specific buffer that is ideal for untreated LAL. Though all LAL reagents produced by FUJIFILM Wako are endotoxin-specific, other commercially available LAL reagents are not. Without this buffer, tests can activate (1,3)-ß-D-glucan when trying to determine the endotoxin concentration in the sample.Endotoxin Extracting Solution
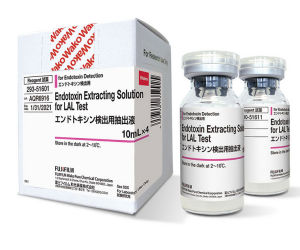
Pharmaceutical
Traditionally, water or saline solution has been used to extract endotoxin in tests involving medical devices and equipment; however, the efficacy of this extraction method is debated. In order to provide our customers with a more reliable method, FUJIFILM Wako has developed a solution containing human serum albumin (HSA) that is capable of extracting endotoxins that cannot be extracted in water or saline solution.Limulus PS Single Test
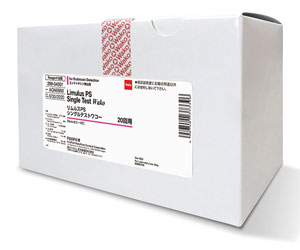
Pharmaceutical
The Limulus PS Single Test contains endotoxin-specific single-test vials and an affinity resin suspension called PyroSep™, which is designed to overcome any product interference by adsorbing potential endotoxin in samples while washing away the inhibitory components.Pyrostar™ ES-F/Plate
Pharmaceutical
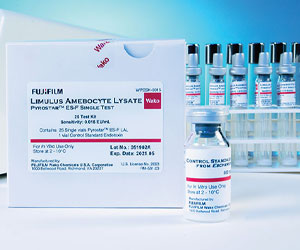
PYROSTAR™ ES-F/PLATE is a multi-test vial kit having the same endotoxin-specific formulation as our other ES-F series products yet designed to be used with the microplate reader. To use this configuration, the user dispenses 0.05 mL of the sample into the microplate followed by 0.05 mL of dissolved LAL reagent.Pyrostar™ ES-F
Pharmaceutical
The PYROSTAR™ ES-F series of reagents are endotoxin-specific and specially formulated to be unreactive to (1,3)-ß-D-glucan. In addition, these products are dual-purpose, meaning they are formulated to be used as either a gel clot or kinetic turbidimetric assay. The kits are available in a single-test or multi-test configuration and are matched with a specific Control Standard Endotoxin (CSE). PYROSTAR™ ES-F reagents work well with samples that have color or with samples that present difficulties in quantification by the KCA. Single-test vials come with pre-dispensed LAL reagent for a single measurement. They are designed to be used with our Toxinometer® Measurement System (Toxinometer®) to assist labs transitioning from a gel clot to a kinetic turbidimetric methodology and is ideal to assay a small number of samples or for a less-experienced user. The multi-test vial is designed to be utilized with a water bath, heat block, or the Toxinometer® and is ideal for a user testing a larger number of samples.Limulus Color KY
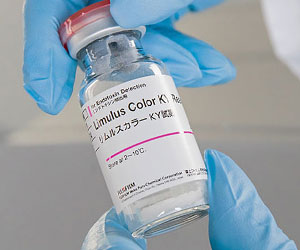
Pharmaceutical
The Limulus Color KY Series are quantitative kinetic chromogenic assays (KCA) that are endotoxin-specific and unreactive to (1,3)-ß-D-glucan. This series includes both a multi-test kit and a single-test kit, which utilize a synthetic substrate that produces a yellow color to detect endotoxin with high sensitivity. All reagent kits are matched with a Control Standard Endotoxin (CSE). These reagents work well with samples that are very turbid or show difficulty in quantification by the kinetic turbidimetric assay (KTA). Limulus Color KY single-test vials come with pre-dispensed LAL reagent for a single measurement. This single test vial is ideal for a user testing a smaller number of samples or one who is less experienced. The multi-test vial is designed to be used with the Toxinometer® or a microplate reader and is ideal for a user with a larger number of samples that are turbid.LAL Reagent Products
Pharmaceutical
FUJIFILM Wako Chemicals U.S.A. Corporation (FUJIFILM Wako) is recognized around the world as a trusted supplier of pure chemicals and reagents. After establishing a sales office in Dallas, Texas, in 1981, we grew significantly in the U.S. and in 1989, relocated our corporate headquarters and manufacturing facility to Richmond, Virginia. In 2012, we culminated three decades of research and development by introducing our PYROSTAR™ ES-F series to the market. The PYROSTAR™ ES-F series offers a simple, accurate platform for the detection of bacterial endotoxin. This endotoxin-specific LAL reagent is accompanied by a suite of complementary products to offer a complete solution for your endotoxin testing needs. Like all FUJIFILM Wako businesses, our LAL division is committed to manufacturing the highest-quality chemical reagents and supplies. Our commitment to delivering exceptional customer service is just as strong. When partnering with our LAL division, you receive personal attention from a team of specialists dedicated to your success. We look forward to providing you with superior service and supporting your laboratory in maintaining the utmost level of quality control Download the FUJIFILM Wako LAL Reagent CatalogMicrobiology Product Areas
Pharmaceutical | ||
| Endotoxin | Show all suppliers | |
| Pyrogen Free Labware | Show all suppliers | |