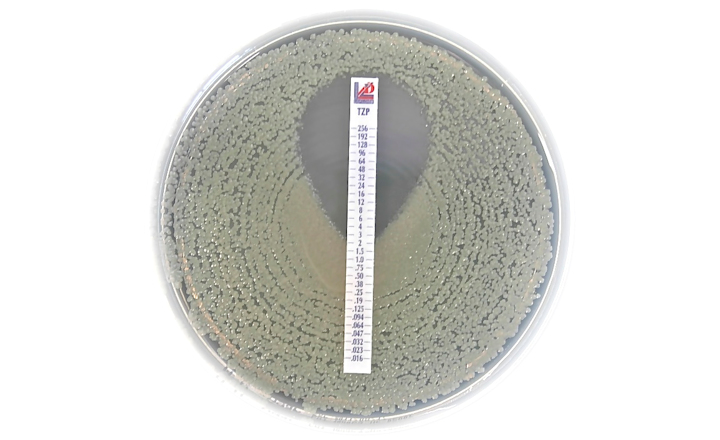
Liofilchem has received clearance from the FDA to market the Piperacillin-tazobactam 0.016/4-256/4 MTS™ (MIC Test Strip) in the United States, a patented quantitative assay for determining the Minimum Inhibitory Concentration (MIC) of piperacillin-tazobactam.
MTS™ TZP can be used to determine the MIC of piperacillin-tazobactam against the following microorganisms for which piperacillin-tazobactam has been shown to be active clinically and/or in vitro according to the FDA drug approved label:
Enterobacterales, Pseudomonas aeruginosa, Acinetobacter baumannii
Ref. 921081 - 10/pack
Ref. 92108 - 30/pack
Ref. 921080 - 100/pack
The entire MTS™ product catalog is CE marked and fully available as IVD for clinical diagnostics purposes in Europe.
The MTS™ range is also compliant with MDSAP (Australia TGA, Brazil ANVISA, Health Canada, USA FDA, Japan MHLW/PMDA) and is registered at the competent Authority in many countries outside Europe as a clinical diagnostic device.
Click here for further information on US FDA-cleared MTS™ items or use the Request Information button below.