Key Points:
- Rapid, point of care tests for SARS-CoV-2 antigen, using saliva or swab samples are now widely available
- These POC tests are less sensitive than the gold-standard RT-PCR but do have high specificity
- Easy to use and interpret, time to result can be between 10 minutes to 40 minutes depending on the type of assay
- Isothermal amplification can be combined with other detection methods, i.e. CRISPR, to improve accuracy.
- Kit manufacturers performance claims are often generated from high viral load samples that may not be representative of real-life samples.
COVID-19 Antigen Detection Tests for Screening
Antigen detection kits on the market target SARS-CoV-2 protein antigens, mainly the nucleoprotein (NP) antigen. There are several different types of antigen detection kits:
- chemiluminescence immunoassay (CLIA)
- fluorescent (FIA) immunoassays
- lateral flow immunoassays (LFIA)
- lateral flow fluorescent immunoassays
Introduction: The World Health Organization (WHO) has declared that rapid antigen detection tests (RADTs) should only be used if RT-PCR is not available and that they must have ≥ 90% sensitivity and 99% specificity. This precautionary approach comes after many reports of false negatives with the first generation of COVID-19 RADTs.
There are also reports of false positives from RADTs, most likely due to the embedded antibodies cross-reacting with other coronaviruses in circulation. These false positives are mainly associated with tests that target the N-protein (nucleocapsid protein); a highly conserved antigen among other human coronaviruses.
However, the spike protein (a component in many COVID-19 vaccines) has a subunit (S1) highly conserved only in SARS-CoV-2. Any antigen test that targets this protein is less likely to give false positives. There is the matter of variants, where mutations occur in the spike protein, which has the potential to invalidate these tests. Testing laboratories should check with the manufacturer if the test has been validated for each variant.
There are several RADTs that can detect SARS-CoV-2 in saliva samples, which is a preferred choice, as it more patient-friendly, but their accuracy can vary.
Automated test readers can improve the sensitivity of most RADTs. They can also digitally record test results, facilitating international travel, and also send a report to public health agencies for surveillance purposes.
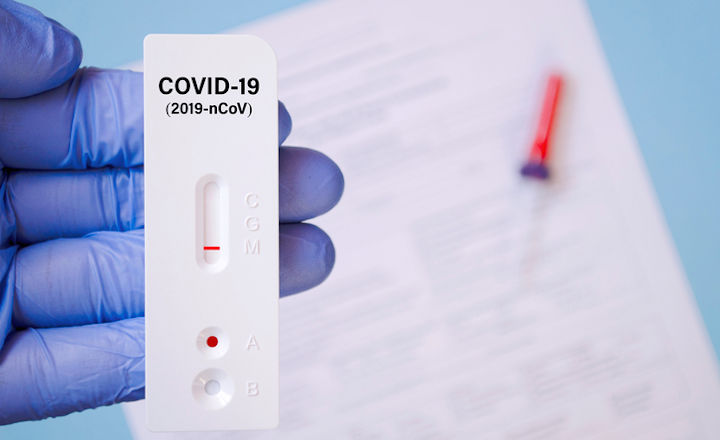
Immunochromatographic/Lateral-Flow Immunoassays (LFIA):
This is based on the design of the common pregnancy test. The test consists of specially-coated antibodies with negative and positive controls immobilized on its strip. The test must be viewed at a specified time-point to read the correct result.
There are also commercialized automatic reading devices, that the diagnostic strip is inserted into, thus avoiding human error. These readers have performed better in validation studies than visual reading. These readers can give a semi-quantitative or quantitative result. These tests can be performed by unskilled operators in near-patient settings and turnaround times are usually 15 minutes. This makes them ideal for ports but only if passenger levels are reduced, as there is a chance of false negatives due to low viral load in the patient not been detected.
COVID-19 Nucleic-Acid/Molecular Near-Patient Testing/Point-of-Care Testing:
Isothermal amplification:
COVID-19 tests using isothermal nucleic acid amplification technology are widely available and are more accurate than lateral-flow tests. Loop-mediated isothermal amplification (LAMP) is the most well-known version of this molecular tool, due to its use during the Zika virus epidemic. LAMP testing does not require thermocycler, and only requires a simple heat-block to provide a constant temperature (ranging from (60-65°C) to activate its two enzymes, one for converting RNA to DNA and another to copy DNA. A set of primers is also added to bind to the targets on the viral genome. 'Loop' structures are formed that allow rapid interpretation for a result (i.e.changes in turbidity, color or fluorescence). This method has a low limit of detection (LoD), but studies have shown it to be less accurate than RT-PCR.
Other variations of this technology include nicking enzyme-assisted reaction (NEAR), recombinase polymerase amplification (RPA), and Insulated Isothermal Polymerase Chain Reaction (RT-iiPCR).
CRISPR & isothermal amplification:
There is several CRISPR SARS-CoV-2 tests on the market using the isothermal amplification method. Proprietary molecular tools such as SHERLOCK and DETECTR tools that amplify the sequence along with the CRISPR Cas 12/13 system for detection have demonstrated similar results to that of PCR.
Sequencing & Isothermal amplification:
A problem with isothermal amplification techniques is that substances present in the sample might affect how the test is read i.e turbidity. The use of nano-sequencing to read results can increase sensitivity and specificity. An example of this test is the LAMPORE assay developed at Oxford University.
Rapid PCR
Several rapid PCR tests are available (FDA-EUA and CE-IVD) and are designed for near-patient settings, with some already in use at airports. These PCR cyclers have a small footprint and typically weigh 1Kg, they analyze swab samples and automate the nucleic acid extraction and amplification, with only basic skills needed to operate. The average turnaround time to result is 40 minutes.