New England Biolabs® (NEB®) has announced the launch of the SARS-CoV-2 Rapid Colorimetric LAMP Assay Kit, a Research Use Only (RUO) product that can be used for the detection of novel coronavirus, SARS-CoV-2 RNA.
The kit serves as a simple alternative to RT-qPCR and enables visual detection of amplification of SARS-CoV-2 nucleic acid in just 30 minutes.
The kit utilizes loop-mediated isothermal amplification (LAMP) to detect specific segments of the viral genome.
This system provides clear visual detection of amplification, based on the generation of protons and the subsequent drop in pH that occurs from the extensive DNA polymerase activity in a LAMP reaction.
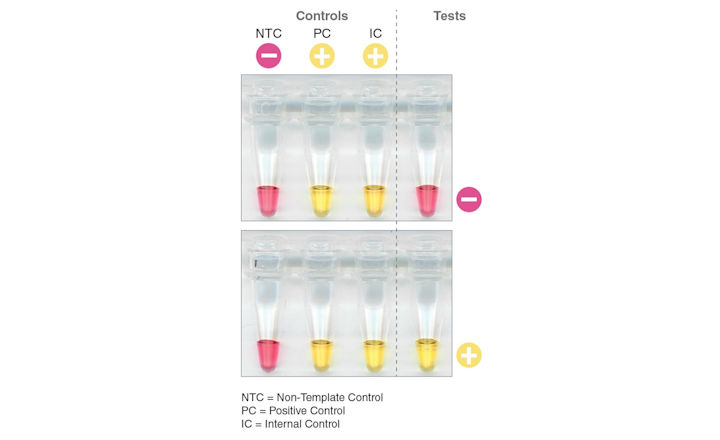
The decrease in pH produces a color change, with pink indicating a negative result and yellow indicating a positive result.
"RT-qPCR, the predominant nucleic acid amplification technology, requires a laboratory with sophisticated equipment, and can take hours to produce a readout," said Steven Chiu, Product Marketing Manager, DNA Amplification at NEB.
"With the SARS-CoV-2 Rapid Colorimetric LAMP Assay Kit, all you need is a simple heat source and 30 minutes to visually detect amplification of SARS CoV-2 RNA. This assay thus has the potential to be very beneficial in a low-resource or point-of-care setting."
The kit includes WarmStart® Colorimetric LAMP 2X Master Mix with UDG (NEB #M1804) and a primer mix targeting the N and E regions of the viral genome.
The WarmStart mix is an optimized formulation of Bst 2.0 WarmStart DNA Polymerase and WarmStart RTx in a special low-buffer reaction solution containing a visible pH indicator for rapid and easy detection of LAMP and RT-LAMP reactions.
The inclusion of dUTP and UDG in the master mix reduces the possibility of carryover contamination between reactions.
Key features of the kit include:
- Constant reaction temperature eliminates the need for sophisticated instrumentation, including thermocyclers
- Assay targets N and E regions of the SARS-CoV-2 genome, enabling optimized sensitivity and specificity
- Includes non-template control, positive control, and internal control to verify assay performance
To learn more about how LAMP is well-suited for point-of-care and field diagnostics, as well as other LAMP assays designed to date, visit here. To learn more about NEB's efforts to support COVID-19 research, visit here.
This RUO product is not approved by the U.S. Food and Drug Administration, or any foreign equivalent, for diagnostic use.
Note: This content has been edited by a rapidmicrobiology staff writer for style and content.