- Tthe Flockscreen LAMP Avian Influenza Molecular Test, is first rapid test to accurately detect the presence of virus in both cloacal and oropharyngeal poultry samples.
- Alveo's portable and rugged molecular diagnostic platform is designed to provide accurate, rapid test results at the point of need.
- Wirelessly upload time-stamped geotagged results to a common portal, providing organizations with near real-time information on the spread of the virus.
Alveo Technologies, Inc. have announced that rigorous development and testing has demonstrated that the Flockscreen LAMP Avian Influenza Molecular Test can accurately detect the presence of virus in both cloacal and oropharyngeal samples from poultry with comparable limit of detection, sensitivity and specificity. Alveo and their development partners performed more than 20,000 reactions on over 4000 assay cartridges during their evaluation of over 100 candidate primer sets and assay conditions, using both contrived and clinical samples. Relevant regulatory bodies have been engaged and appropriate validation and verification activities will be completed prior to shipment.
The product is available today for pre-sale in Europe and the Middle East. The company expects to begin shipping in volume in the third quarter of 2024.
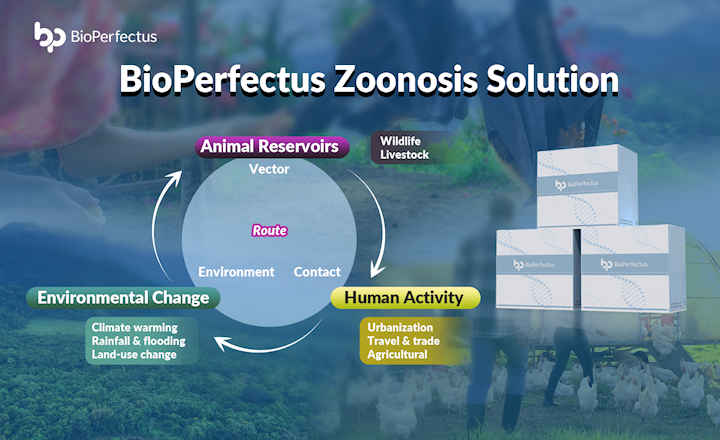
The current HPAI panzootic has recently spread to cattle and at least 47 other mammalian species. Due to current disease control legislation, this has led to the destruction of more than half a billion poultry worldwide. Rapid, accurate tests are vital for early detection and containment, protection of the poultry industry, the prevention of zoonotic transmission and spillover events from one species to another, food security, the maintenance of agricultural trade, and ongoing monitoring and surveillance.
Current agricultural tests for highly pathogenic avian influenza (HPAI) all suffer from one of several significant drawbacks that make them unsuited to these use cases. Polymerase chain reaction (PCR) requires lab processing, which means it can take days to receive a result, and vaccination causes serology tests to turn positive even if the bird is not infected1. Lateral flow (also known as antigen or instant tests) lacks sensitivity and produces a significant percentage of false negative results.
Alveo’s portable and rugged molecular diagnostic platform has been designed to provide accurate, rapid test results at the point of need. It functions as a core component of a notifiable disease electronic management ecosystem that the company and its strategic partners have developed to support producers, as well as governmental bodies who are looking to expand the surveillance of important zoonotic pathogens.
Two multiplexed panels enable the on-site broad detection of Avian Influenza Type A virus (targeting matrix gene) in poultry with differentiation of H5, H7, and H9 subtypes using oropharyngeal or cloacal samples. Both panels contain 7 assays, 6 of which are identical between cartridges. This configuration ensures the greatest coverage of the most clinically relevant global AI strains from the past 5-10 years, through to the current strains.
The platform’s rugged design enables it to function in the field under extreme conditions, including very hot and near freezing temperatures, following a drop of one meter, and when subjected to movement and vibration, which are common challenges encountered in field settings.
Alveo has developed assays for Flockscreen LAMP AI on its proprietary molecular platform with several partners —including x-OvO, Royal GD, and Pharmsure International Ltd. — and it will provide healthcare and government authorities with a rapid, decentralized, molecular test that eliminates the need to ship hazardous samples to a lab. Additionally, the platform has the capability to wirelessly upload time-stamped geotagged results to a common portal, providing organizations with near real-time information on the spread of the virus. The platform will provide veterinarians with intuitive, rapid, point-of-need testing. For producers, Flockscreen will empower rapid responses to reduce the disease’s spread and mitigate the impact on their business.
“Alveo’s molecular diagnostic platform is a groundbreaking technology that raises the bar for speed and accuracy in point-of-need diagnostic testing,” said Wim Kieftenbeld, Director Business Development at Royal GD. “Combined with our expertise in veterinary diseases and diagnostics, we’ve worked together to develop an assay that accurately and rapidly delivers results at point of need for both cloacal and oropharyngeal samples, a first for agricultural diagnostics for HPAI.”
“Our platform makes it simple to test for HPAI in poultry, and we will be the first diagnostic of its kind that can test both cloacal and oropharyngeal samples,” said Shaun Holt, CEO at Alveo. “Our rugged design enables healthcare providers, veterinarians, growers, and public health officials to obtain rapid results at point of need so they can take immediate action.”
Alveo expects first customer shipments of Flockscreen LAMP AI in EMEA to begin by Q3 2024.
For more information visit: alveotechnologies.com/flockscreen
1 Tumpey TM, Alvarez R, Swayne DE, Suarez DL. Diagnostic approach for differentiating infected from vaccinated poultry on the basis of antibodies to NS1, the nonstructural protein of influenza Avirus. J Clin Microbiol. 2005 Feb;43(2):676-83. doi:10.1128/JCM.43.2.676-683.2005. PMID: 15695663; PMCID: PMC548100.