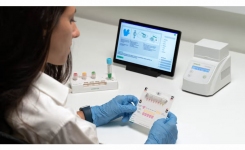
Developed and commercialised the CE marked Dragonfly™ is a <30 minutes sample-to-result in vitro diagnostic test, based on isothermal LAMP technology, intended for the extraction and qualitative detection of SARS-CoV-2 RNA as a single target and combined with the extraction and detection of Influenza A virus, Influenza B virus, human respiratory syncytial virus and human rhinovirus in a multiplex panel from a nasopharyngeal, nasal, throat, and buccal samples.
Microbiology Product Areas
Clinical & Veterinary
Influenza
Show all suppliersLAMP/Isothermal Amplification
Show all suppliersRespiratory Syncytial Virus (RSV)
Show all suppliersRhinovirus
Show all suppliersSARS-CoV-2
Show all suppliersAll Product News
ProtonDx's Dragonfly Evaluated for Identification of Hidden Malaria Cases to Aid Elimination Efforts
Posted: Nov 03, 2025
Posted: Jan 09, 2023
Posted: Sep 02, 2022
Posted: Jul 27, 2022
LATEST MICROBIOLOGY NEWS
MICROBIOLOGY EVENTS
-
PDA Good Aseptic Manufacturing Conference 2026
11 Jun 2026 -
Better Process Control School (Virtual)
16 Jun 2026 -
Drug Discovery USA 2026
16 Jun 2026 -
Biolog Webinar Shows How to Achieve Consistent Anaerobic Culture Results
16 Jun 2026 -
VITAL 4.0 in Practice - Webinar
18 Jun 2026 -
ISO 24914:2026 – A New International Standard for LAMP Technology in Food Safety Testing
23 Jun 2026 -
New Rapid Microbial Testing System with Results in Hours
25 Jun 2026 -
PDA Advanced Therapy Medicinal Products Conference 2026
25 Jun 2026 -
ADLM 2026
26 Jul 2026 -
IAFP 2026
26 Jul 2026