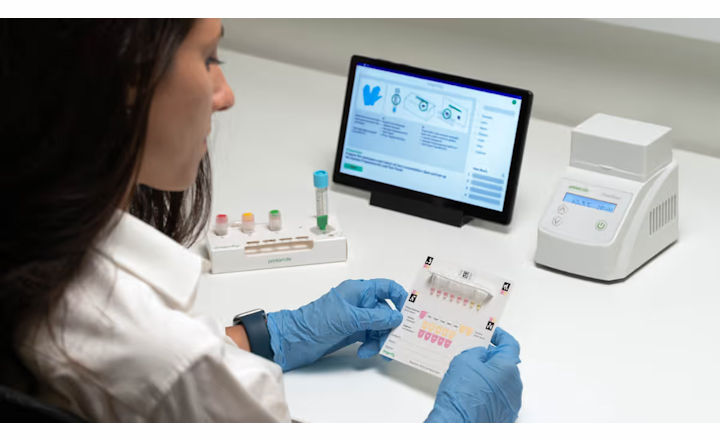
ProtonDx have announced a strategic partnership with innovative commercial POCT provider Katalyst Laboratories, to distribute ProtonDx’s Dragonfly™ rapid in vitro diagnostic system.
Under the terms of the agreement, ProtonDx has granted Katalyst exclusive rights to the distribution of Dragonfly multi-pathogen detection system in a variety of Katalyst client segments. Katalyst has focused on providing disruptive diagnostic solutions that deliver customer-centric services to meet the needs of the rapidly evolving diagnostics landscape, making them an ideal partner for the new Dragonfly system.
Bob Enck, Chairman and President of ProtonDx, stated: “I am pleased to announce our strategic partnership with Katalyst for the distribution of our proven Dragonfly diagnostic system. Katalyst are leading the evolution of in-field diagnostic testing for the film and TV production industries, providing ready access to novel diagnostic solutions to support operational continuity for these people-centric businesses. Dragonfly is a potential game-changer in the diagnostics industry - providing rapid and accurate results, irrespective of location.”
Timely pathogen determination, wherever it is needed, can also help mitigate unnecessary usage of antibiotics and therefore, play a role in helping to address the global challenge of antimicrobial resistance (AMR). Dragonfly enables cost-effective multi-pathogen detection, using proprietary ultra‑fast nucleic acid extraction and isothermal detection, achieving PCR-equivalent sensitivity and specificity in less than 30 minutes from sample to result.
Andrew Wheeler, co-founder and CEO of Katalyst Laboratories, said: “Dragonfly offers robust infectious disease detection and testing capability at the point-of-need. We are excited to be working with the expert team at ProtonDx and distributing this stand-out solution that can deliver high-quality molecular diagnostic testing anywhere. By facilitating robust, accurate and reliable point-of-need testing, Dragonfly complements our comprehensive range of testing solutions. Dragonfly offers the diagnostics community a unique and cost-effective testing solution that can be readily adapted to support the rapidly evolving need for accurate pathogen detection. Something that I believe can add value not only within the film & TV market, but across all point-of-care channels.”
A number of pathogen identification Test Panels are available on the Dragonfly platform, including the 5-in-1 Respiratory Test Panel. Respiratory tract infections often exhibit similar symptoms and thus require accurate diagnostic differentiation to ensure that patients receive appropriate treatment. The multi-pathogen Dragonfly panel is intended to aid in the identification of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Influenza A Virus (IAV), Influenza B Virus (IBV), Respiratory Syncytial Virus (RSV) and Human Rhinovirus (HRV) in samples from patients regardless of gender or age.
Dragonfly’s reliability, ease-of-use, and portability have been validated by its successful deployment at the Winter Olympic Games and the 2022 Commonwealth Games1 in Birmingham, UK, where it was a key part of the diagnostic testing portfolio. This rapid, accurate and robust system is poised to revolutionise rapid infectious disease diagnosis and tracking worldwide.
1 Also known as the XXII Commonwealth Games and Birmingham 2022.