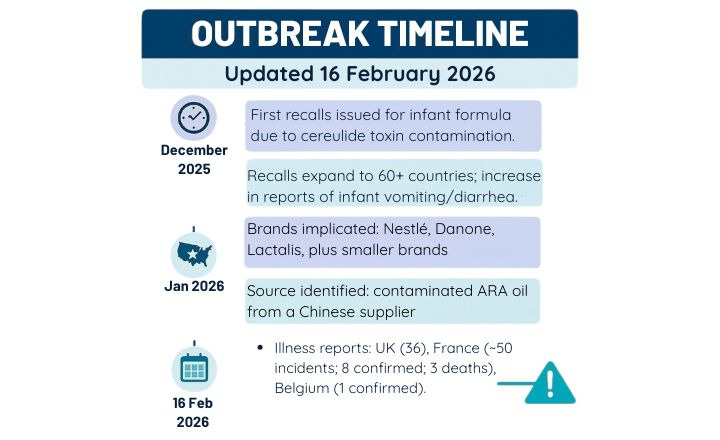
As of February 2026, multiple infant-formula brands sold in the European Union and associated international markets have been recalled or are under investigation due to confirmed or potential contamination with cereulide, a heat-stable toxin produced by Bacillus cereus linked to food poisoning symptoms in infants (e.g., vomiting, diarrhea, dehydration). The recalls began in early December 2025 and have continued into January–February 2026, affecting products sold in 60+ countries1,2.
There are a growing number of reports of diarrhea in infants following consumption of the recalled products. In the UK, there are 36 clinical reports of children developing symptoms consistent with toxin poisoning3, while French authorities reported about 50 “incidents” including 8 confirmed cases and three infant deaths4. A single case in Belgium has been confirmed5.
Brands implicated or recalled due to potential or confirmed cereulide contamination currently include: Nestlé Group, Danone, and Lactalis1,6. Several smaller brands e.g. Vitagermine and Hochdorf Swiss Nutrition formulas have also been recalled as a precautionary action6.
The source of the contamination in this event has been traced primarily to cereulide in arachidonic acid (ARA) oil, a raw ingredient supplied by a third party and used widely in premium infant-formula formulations1,7. As well, base powder to which ARA oil had been added7. Investigations by national authorities and industry reporting identified that this ARA oil came from a Chinese supplier, widely reported to be Cabio Biotech based in Wuhan, China2,5. This supplier’s ARA products were used by multiple infant-formula manufacturers including Nestlé and Danone2,8.
Unlike Salmonella spp. and Cronobacter sakazakii, which are explicitly regulated in powdered infant formula under Commission Regulation (EC) No 2073/2005 on microbiological criteria for foodstuffs, cereulide itself is not currently assigned a specific maximum limit in EU legislation. EU microbiological criteria for Bacillus cereus are risk-based and organism-focused rather than toxin-specific. While some Member States apply guideline values for B. cereus counts in foods (often 10³–10⁵ CFU/g depending on food category), there is no harmonized EU maximum level for cereulide toxin in infant formula.
This regulatory gap is significant because:
- Cereulide is pre-formed and heat-stable
- It survives pasteurization and spray-drying
- Its presence does not necessarily correlate with viable B. cereus counts at the time of testing
Thus, finished-product compliance testing based solely on organism enumeration may fail to detect toxin risk.
References:
-
European Centre for Disease Prevention and Control (ECDC). Precautionary global recall of infant nutrition products following detection of Bacillus cereus toxin (cereulide). Stockholm: ECDC; 2026. Accessed on Feb 12, 2026
-
Reuters. Danone recalls certain baby formula batches produced in Ireland as toxin scare widens. Reuters News Service. January 2026. Accessed on Feb 12, 2026
-
The Standard. UKHSA reports 36 notifications of baby formula cereulide poisoning. February, 2026. Accessed on Feb 12, 2026
-
RTL. February, 2026. Laits infantiles: le ministère de la Santé annonce la mort d'un troisième bébé ayant consommé un produit rappelé en France. Accessed on Feb 12, 2026
-
Euronews. February, 2026. Baby formula recalls: UK investigates 36 babies with suspected cereulide intoxication. Accessed on Feb 1, 2026
-
Euronews. Why baby formula is being recalled across Europe: toxin contamination puts companies on alert. Euronews Health. January 2026. Accessed on Feb 12, 2026
-
Food Safety Authority of Ireland (FSAI). Recall of specific batches of Aptamil and Cow & Gate infant formula due to possible presence of cereulide toxin. Dublin: FSAI; February 2026. Accessed on Feb 12, 2026
-
Yicai Global. Nestlé’s Chinese ARA supplier plunges after Swiss firm issues global recall of baby formula. January 2026. Accessed on Feb 12, 2026