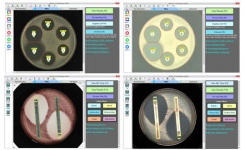
Giles Scientific's recent software updates will help your lab stay in line with regulatory guidelines for antimicrobial susceptibility testing. Allowing your team to focus on getting accurate results because the software takes care of the complicated part of following the rules.
The following guidelines are now available in the 2026 BIOMIC V3 software release:
CLSI (Clinical Laboratory Standards Institute) Guidelines:
- M100, 36th Edition: Performance Standards for Antimicrobial Susceptibility Testing (January 2026)
EUCAST (European Committee on Antimicrobial Susceptibility Testing) Guidelines:
- Breakpoint Tables for Interpretation of MICs and Zone Diameters v 16.0 (January 2026)
- Routine and extended internal quality control for MIC determination and disk diffusion as recommended by EUCAST. v 16.0 (January 2026)
- Zone diameter breakpoints for rapid antimicrobial susceptibility testing (RAST) directly from blood culture bottles. v 8.1 (July 2025)
Please note : Any products described on this page are
for Research Use Only and not intended for clinical diagnostic procedures unless otherwise stated.