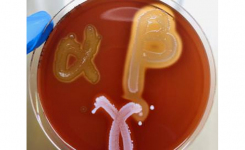
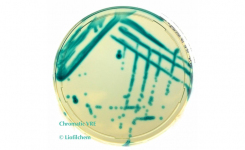
At Liofilchem we are proud of our long-term compliance with the main quality standards in the microbiology industry.
Liofilchem has obtained the EU Quality Management System Certificate (IVDR) pursuant to Regulation (EU) 2017/746 on in-vitro Diagnostic Medical Devices, Annex IX Chapters I and III (Class C and B Devices excluding self/near-patient-testing and Companion Diagnostics). Download here
Liofilchem is certified by TÜV Süd to ISO 13485 as manufacturer of IVD (in-vitro diagnostics medical device) and to the MDSAP scheme (Medical Device Single Audit Program) for the sale of IVD (in-vitro diagnostic medical devices) in Australia, Brazil, Canada, Japan and the United States. Download here
Since 1997 the implementation of the Quality System Management (UNI EN ISO 9001:2015) has allowed us to satisfy Customer expectations, optimize resources, qualify and consolidate the company position in national and international market. Download here
Liofilchem has implemented an “Occupational Health and Safety Management System” which fullfils the requirements of the standard ISO 45001, to control risks in this regard and to improve its performance. Download here
Visit Liofilchem for more or use the Request Information button below to email the company directly.