Commercially manufactured antibody tests check for SARS-CoV-2 antibodies in individuals and are available through healthcare providers and commercial laboratories. CDC is evaluating the performance of these tests in collaboration with the following federal organizations:
- Biomedical Advanced Research and Development Authority
- U.S. Food and Drug Administration (FDA)
- National Institutes of Health
- Department of Defense
- White House Office of Science and Technology Policy
Concerning performance results, please note: These estimates of sensitivity and specificity are just that: estimates. They include 95% confidence intervals, which are the range of estimates we are about 95% sure a test's sensitivity and specificity will fall within given how many samples were used in the performance validation.
The more samples used to validate a test, the smaller the confidence interval becomes, meaning that we can be more confident in the estimates of sensitivity and specificity provided.
Tests are also described by their Positive and Negative Predictive Values (PPV and NPV). These measures are calculated using a test's sensitivity, its specificity, and using an assumption about the percentage of individuals in the population who have antibodies to SARS-CoV-2 (which is called "prevalence" in these calculations).
Every test returns some false positive and false-negative results. The PPV and NPV help those who are interpreting these tests understand, given how prevalent individuals with antibodies are in a population, how likely it is that a person who receives a positive result from a test truly does have antibodies to SARS-CoV-2 and how likely it is that a person who receives a negative result from a test truly does not have antibodies to SARS-CoV-2.
The PPV and NPV of a test depend heavily on the prevalence of what that test is intended to detect. Because all tests will return some false positive and some false-negative results, including tests that detect antibodies to SARS-CoV-2, broad use of the tests, when not appropriately informed by other relevant information, such as clinical history or diagnostic test results, could identify too many false-positive individuals.
How performance measures were calculated: In some cases, there may be minor differences between the numbers on this page and those in the instructions for use. Confidence intervals for sensitivity and specificity that appear on this page were calculated per a score method described in CLSI EP12-A2 (2008). See Recognized Consensus Standards. Confidence intervals for PPV and NPV were calculated using the values from the 95% confidence intervals for sensitivity and specificity. (FDA)
Test Performance
Abbott Alinity i SARS-CoV-2 IgG
Developer: Abbott
Test: Alinity i SARS-CoV-2 IgG
Technology: High Throughput CMIA
Target: Nucleocapsid
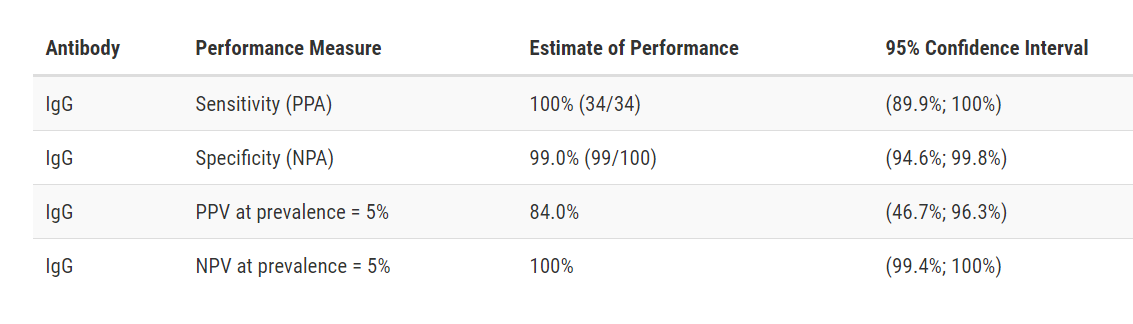
Test Facts:
Information for Healthcare Providers
Abbott Architect SARS-CoV-2 IgG
Developer: Abbott
Test: Architect SARS-CoV-2 IgG
Technology: High Throughput CMIA
Target: Nucleocapsid
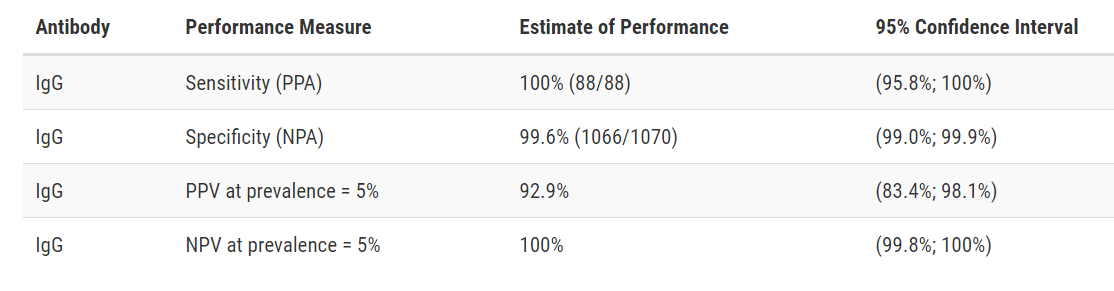
Test Facts:
Information for Healthcare Providers
Autobio Anti-SARS-CoV-2 Rapid Test
Developer: Autobio
Test: Anti-SARS-CoV-2 Rapid Test
Technology: Lateral Flow
Target: Spike
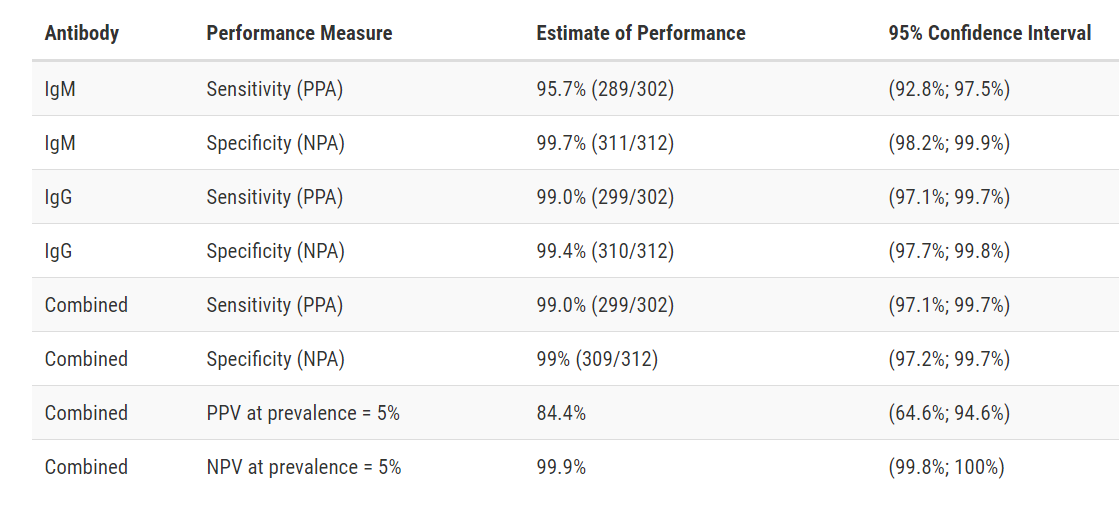
Test Facts:
Information for Healthcare Providers
Bio-Rad Platelia SARS-CoV-2 Total Ab
Developer: Bio-Rad Laboratories, Inc
Test: Platelia SARS-CoV-2 Total Ab
Technology: ELISA
Target: Nucleocapsid
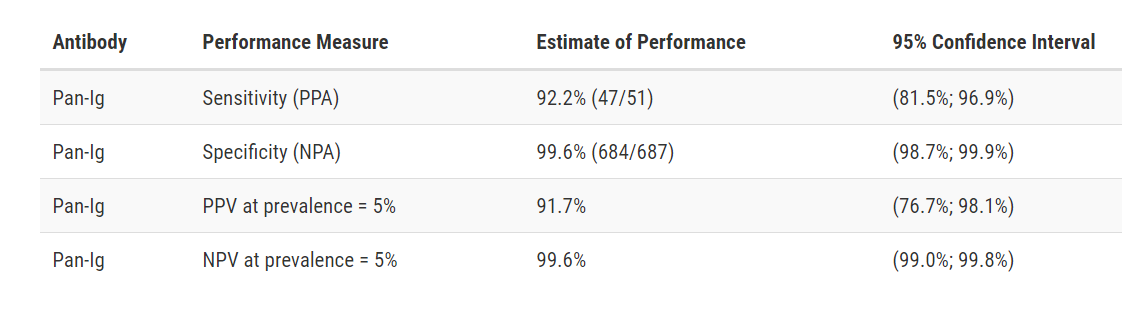
Test Facts:
Information for Recipients
Information for Healthcare Providers
Cellex qSARS-CoV-2 IgG/IgM Rapid Test
Developer: Cellex, Inc.
Test: qSARS-CoV-2 IgG/IgM Rapid Test
Technology: Lateral Flow
Target: Spike and Nucleocapsid
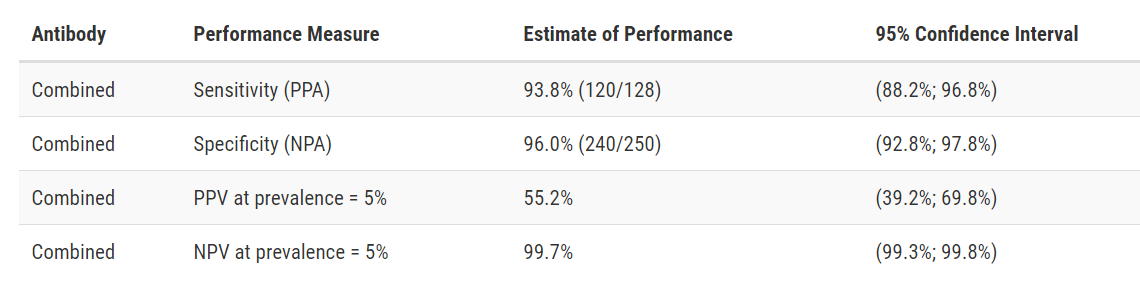
Test Facts:
Information for Healthcare Providers
Chembio Diagnostic Systems DPP Covid-19 IgM/IgG System
Developer: Chembio Diagnostic Systems, Inc.
Test: DPP Covid-19 IgM/IgG System
Technology: Lateral Flow with Reader
Target: Nucleocapsid
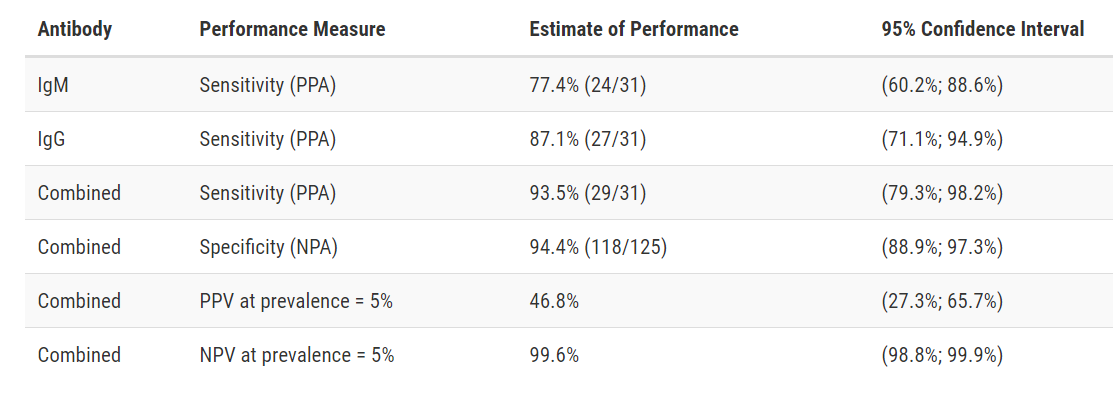
Test Facts:
Information for Healthcare Providers
DiaSorin LIAISON SARS-CoV-2 S1/S2 IgG
Developer: DiaSorin
Test: LIAISON SARS-CoV-2 S1/S2 IgG
Technology: High Throughput CMIA
Target: Spike
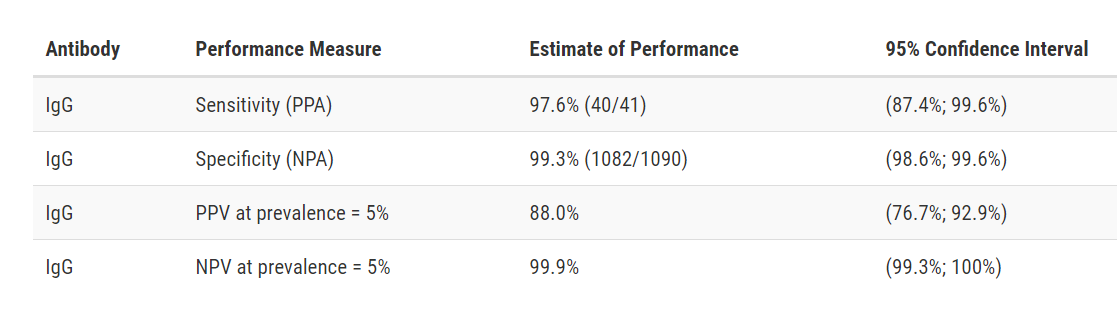
Test Facts:
Information for Healthcare Providers
Developer: EUROIMMUN
Test: SARS-COV-2 ELISA (IgG)
Technology: ELISA
Target: Spike
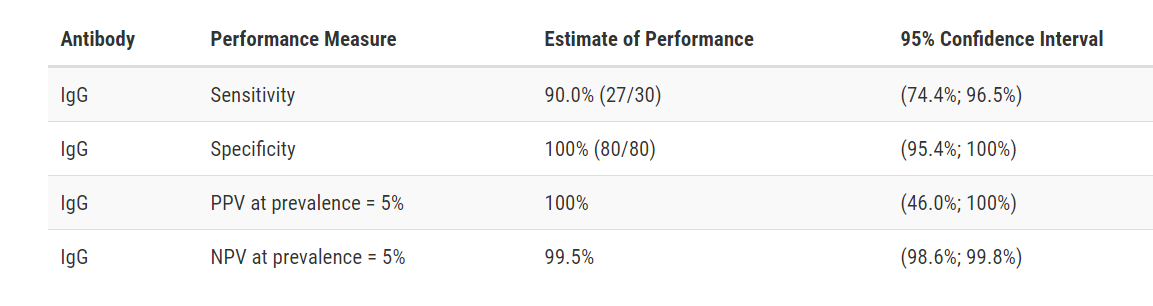
Test Facts:
Information for Healthcare Providers
NCI's Frederick National Laboratory for Cancer Research Evaluation Report
NCI's Frederick National Laboratory for Cancer Research Evaluation Data
Healgen COVID-19 IgG/IgM Rapid Test Cassette
Developer: Healgen
Test: COVID-19 IgG/IgM Rapid Test Cassette
Technology: Lateral Flow
Target: Spike
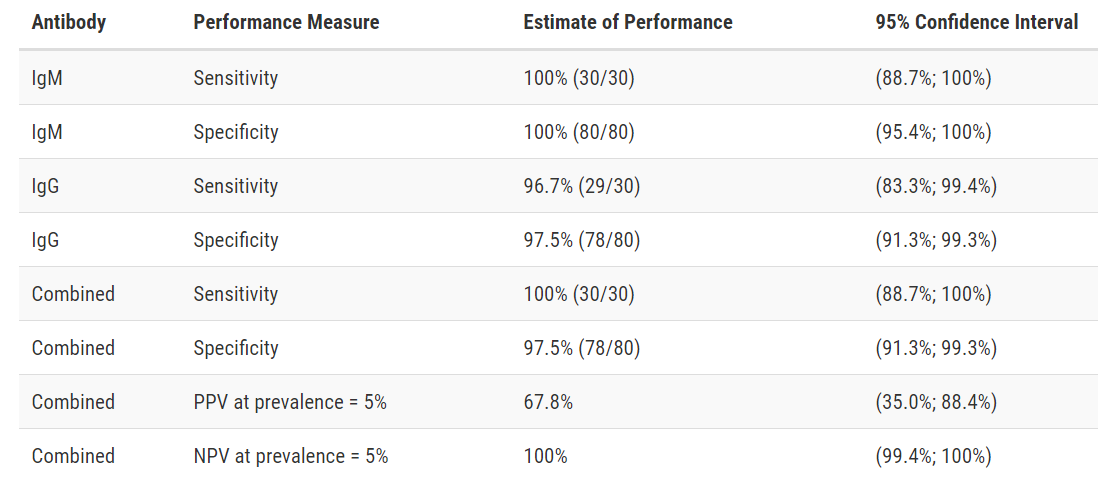
Test Facts:
Information for Healthcare Providers
NCI's Frederick National Laboratory for Cancer Research Evaluation Report
NCI's Frederick National Laboratory for Cancer Research Evaluation Data
Mount Sinai Hospital Clinical Laboratory COVID-19 ELISA Antibody Test
Developer: Mount Sinai Hospital Clinical Laboratory
Test: Mt. Sinai Laboratory COVID-19 ELISA Antibody Test
Technology: 2-Step ELISA
Target: Spike
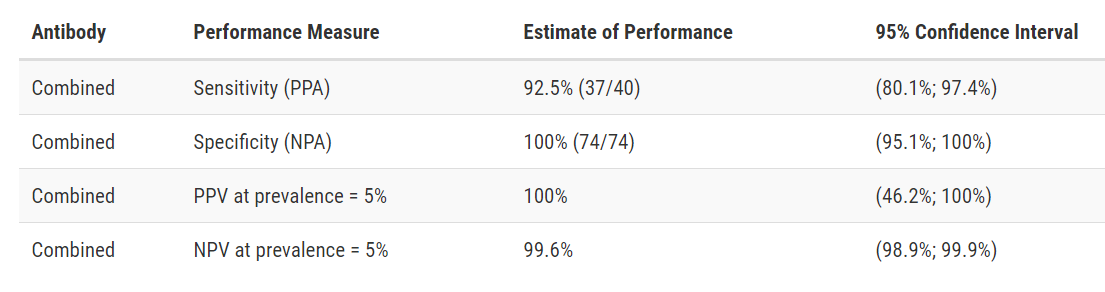
Test Facts:
Information for Healthcare Providers
Ortho-Clinical Diagnostics VITROS Anti-SARS-CoV-2 IgG test
Developer: Ortho-Clinical Diagnostics, Inc.
Test: VITROS Anti-SARS-CoV-2 IgG test
Technology: High Throughput CLIA
Target: Spike
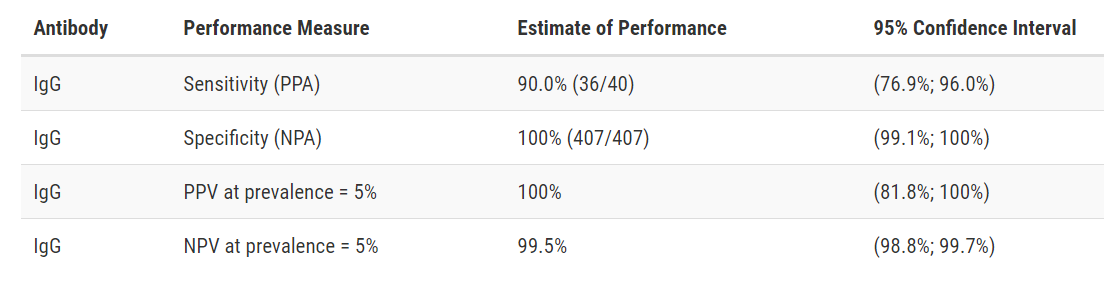
Test Facts:
Information for Healthcare Providers
Developer: Ortho-Clinical Diagnostics, Inc.
Test: VITROS Immunodiagnostic Products Anti-SARS-CoV-2 Total Reagent Pack and Calibrator
Technology: High Throughput CLIA
Target: Spike
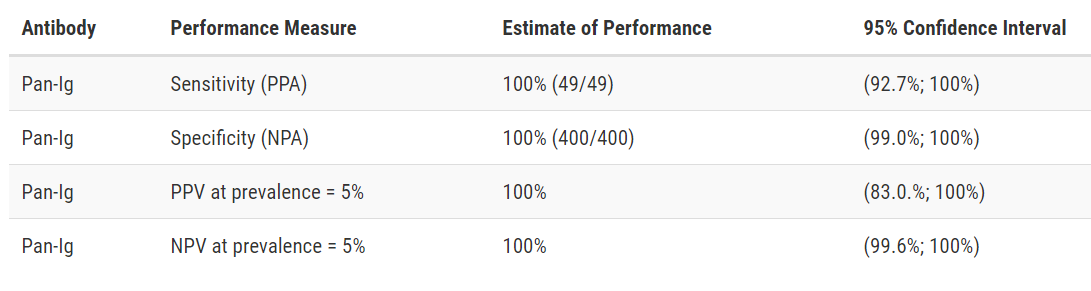
Test Facts:
Information for Healthcare Providers
Developer: Roche
Test: Elecsys Anti-SARS-CoV-2
Technology: High Throughput ECLIA
Target: Nucleocapsid
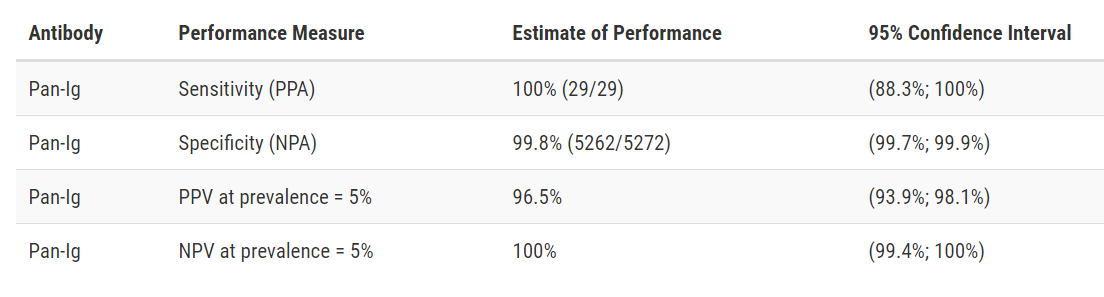
Test Facts:
Information for Healthcare Providers
Siemens Healthcare Diagnostics ADVIA Centaur SARS-CoV-2 Total (COV2T)
Developer: Siemens Healthcare Diagnostics
Test: ADVIA Centaur SARS-CoV-2 Total (COV2T)
Technology: High Throughput CMIA
Target: Spike
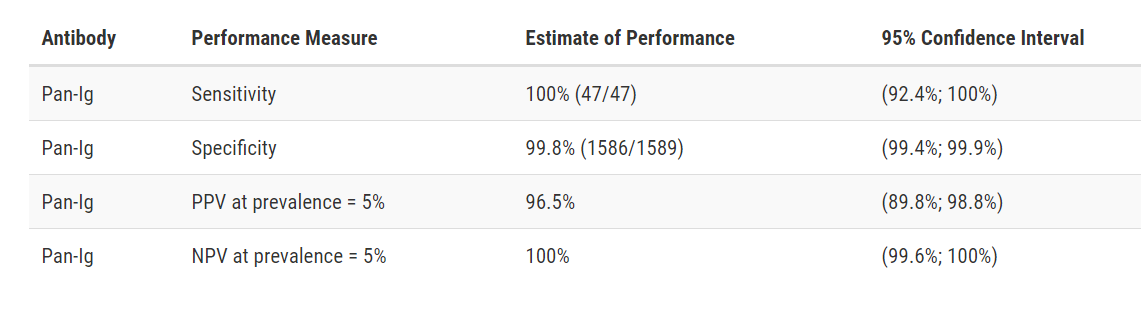
Test Facts:
Information for Healthcare Providers
Siemens Healthcare Diagnostics Atellica IM SARS-CoV-2 Total (COV2T)
Developer: Siemens Healthcare Diagnostics
Test: Atellica IM SARS-CoV-2 Total (COV2T)
Technology: High Throughput CMIA
Target: Spike
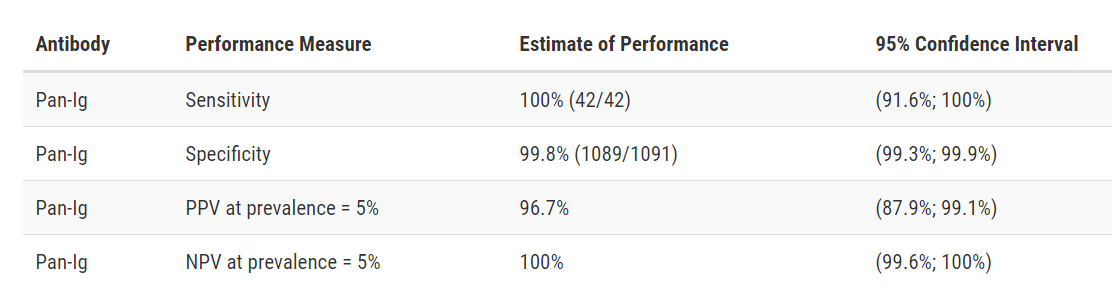
Test Facts:
Information for Healthcare Providers
Wadsworth New York SARS-CoV Microsphere Immunoassay for Antibody Detection
Developer: Wadsworth Center, New York State Department of Health
Test: New York SARS-CoV Microsphere Immunoassay for Antibody Detection
Technology: MIA
Target: Nucleocapsid
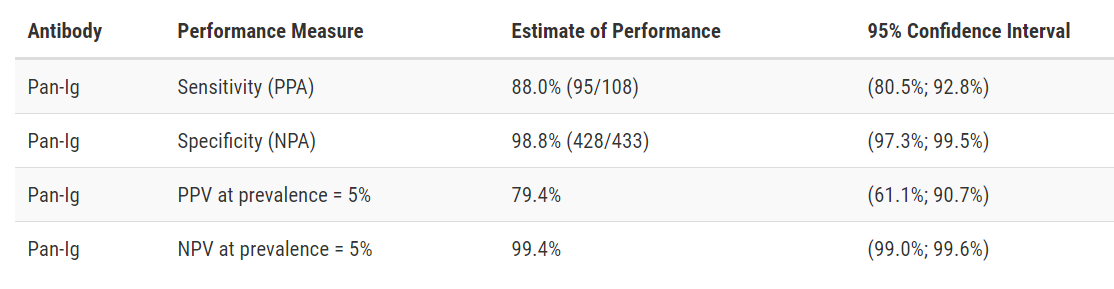
Test Facts:
Information for Healthcare Providers
**The content on this page is valid as of the 9 June 2020, for further updates please click here***