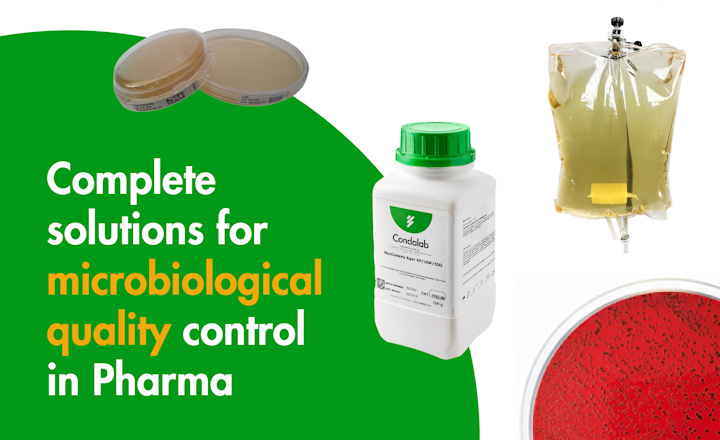
In microbiology, culture media are the gold standard, therefore a large majority of microbiology analysis depends on the quality of culture media and their ability to provide reliable, consistent, and reproducible results in any laboratory.
To address this situation, ISO 11333 has been adopted by users and manufacturers for the quality assurance control of culture media used for food and water analysis. The performance testing of the culture media will depend on whether you are a user or a manufacturer, as well as if it is a ready-to-use media or dehydrated culture media (DCM).
If you are a user of ready-to-use media, the testing will be quite simple as long as the manufacturer has a quality program in place, as with Condalab. All our media performance data is reflected in the easily available documentation, certificates of analysis (CoA), and technical data sheets (TDS), and as a result, our customers don’t have to carry out an exhaustive analysis of the media.
In the case of prepared media from dehydrated formulas (DCM), the user will have to carry out tests depending on the use of the medium (count, enrichment, confirmation), because, even though Condalab also supplies a certificate of quality for DCM, the preparation has a direct impact on the media performance.
Condalab follows ISO 11333:2014, and its amendments A.1:2018 and A.2:2020 to assess the productivity, selectivity, and specificity of our culture media to assure our users:
- every batch of medium is acceptable,
- every medium is fit for purpose, and
- every medium performs accordingly
Watch our CondalabTalk about the Importance of ISO 11333:2014 in QC labs for more information.
If you would like further information, visit Condalab, where an expert team will be glad to support you, or click on the Request Information button below.