A Review of Released BIOBALL MultiShot 550 Batches from 2014 to 2018
Article by Brendan Tindall, Global Solutions Manager – BIOBALL
Unlike our counterparts in the chemical world, consistently precise and accurate results from microbiological quality control are encountered less often. In other words, quantitative Microbiological testing allows for (fairly large) discrepancies in the results due to the inherent difficulties in dealing with living organisms as well as quantifying them. The intellectual properties and proprietary methodologies used in making BIOBALL has redefined what was considered as Quantitative Microbiology and has made it simple to get precise & accurate quantitative microbiological quality controls batch to batch. BIOBALL was developed in Sydney Australia in 2001 and the business was purchased by bioMerieux in 2007. The product continues to be manufactured in Sydney Australia and is now available for purchase all over the world, with millions of tests conducted every year.
SUMMARY
The internal Quality Control results of BIOBALL MultiShot 550 batches released from the beginning of 2014 to 2018 were compiled in this review, all results summarized herein were well within the established specifications. The results from accelerated stability testing and annual real time testing at 13 and 25 months were also included. These results indicate that a mean of 45 - 55 CFU per aliquot could be maintained for all investigated batches to the end of expected shelf life.
Product Introduction
The BIOBALL product is a small freeze dried water soluble ball containing a precise number of viable microorganisms. The manufacturing process uses patented technology and proprietary techniques for precision and viability. A flow cytometer is used to select individual cells from a culture and dispense them into a single droplet. Each droplet therefore contains a precise number of viable cells that have been individually selected for maximum recovery rate on non-selective media. This technology is capable of distinguishing between single cells and cell clusters in cultures such as Staphylococcus aureus. Droplets are frozen in liquid nitrogen, dispensed in vials, freeze-dried and sealed under vacuum for maximum shelf-life and recovery.
BIOBALL is manufactured in Sydney, Australia and produced to the world's highest quality standards, based on ISO:17034 accreditation. The BIOBALL manufacturing site, which is accredited as a Reference Materials producer, was the first company accredited for quantitative microbiological reference standards in the world. ISO:17034 accreditation is issued by NATA, Australia's government endorsed provider of accreditation for laboratories and similar testing facilities. In 2018 BIOBALL available strains products have all been accredited as "Certified Reference Materials".
The BIOBALL comes in four distinct quantitative ranges, SingleShot, MultiShot 550, HighDose 10K and MultiShot 10e8. The SingleShot product is designed for single use. The MultiShot 550 is designed specifically to optimise growth promotion testing workflows. The 550 product is designed to dispense 10, 100μL doses of 50 CFU with minimal variance between results. The HighDose product is generally used for large high-dose inoculations. Finally, the MultiShot 10e8 product is designed for Antimicrobial/Preservative Effectiveness testing.
Due to the unique manufacturing process for BIOBALL every batch has a specific target mean CFU number (not range) and has very tight quality control release requirements. Graphing of the last four years of MultiShot 550 batches shows minimal batch-to-batch variability with respect to time. Each batch of BIOBALL listed below was recovered on different nutrient agar batches strongly suggesting the results are independent of media batches.
Microorganisms
The BIOBALL MultiShot 550 has 11 microorganisms in its range including bacteria, yeast and mould. These include the 5 major strains for GPT in Microbial Enumeration Tests USP <61> and 2.6.12: Aspergillus brasiliensis, Bacillus subtilis, Candida albicans, Pseudomonas aeruginosa and Staphylococcus aureus. As well as the additional strains from USP <71> and 2.6.13 test for Specified Microorganisms: Clostridium sporogenes, Escherichia coli and Salmonella abony. Enterococcus faecalis and Propionibacterium acnes were added to the product line per market request. Finally included is Burkholderia cepacia which currently is not required in any pharmacopoeias yet but is included in the Latest USP revisions from the USP Microbiology expert committee. Also the FDA has stated that they will not release any new products without specific Burkholderia cepacia testing. (https://www.fda.gov/Drugs/DrugSafety/ucm559508.htm )
This study will focus on the results from the 5 major strain for GTP (Aspergillus brasiliensis, Bacillus subtilis, Candida albicans, Pseudomonas aeruginosa and Staphylococcus aureus) plus the Burkholderia cepacia strain.
Product testing
The mean and standard deviation of each batch is determined by a minimum of 50 counts(i.e. aliquots counted on a singular plate of the relevant agar type) per batch following internal standard operating procedures. The mean CFU per Bioball target is 550±50 CFU for the MultiShot 550 product and the standard deviation target is ≤10% the product mean.
The standard operating procedure for the quality control plating of the various batch mimics the “Instructions for Use” (detailed in the relevant instruction manual sent with each product). All plating and counting is done manually with dedicated and experience staff. Details of the agar plates used and incubation conditions for each organism are detailed in Table 1.
Table 1: List of the different organisms offered in the 550 format
| Organism |
Recommended Agar Culture Media |
Recommended Incubation Conditions |
Cell Culture Preparation Type |
Number of Released Batches (2014 - 2018) |
| Aspergillus brasiliensis | DRBC agar | 37o C | Spore | 83 |
| Bacillus subtilis | Nutrient agar | 37o C | Spore | 87 |
| Burkholderia cepacia | Nutrient agar | 37oC | Cell | 18 |
| Candida albicans | Nutrient agar | 37oC | Cell | 90 |
| Pseudomonas aeruginosa | Nutrient agar | 37oC | Cell | 98 |
| Staphylococcus aureus | Nutrient agar | 37oC | Cell | 96 |
Results – Quality control results
Figure 1 through 6 illustrate the quality control results of all the released batches for the 6 investigated strains. It can be seen that all the means are well within the expected range. Further sections will analyse the batches stability and moving range.
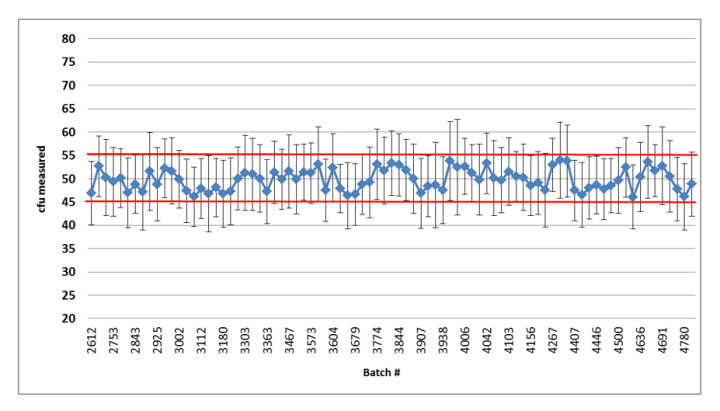
Figure 1: Summary graph of the quality control results of the released Bioball 550 batches of Aspergillus brasiliensis: error bars represent standard deviation based on the repeated aliquots; Red lines indicate accepted mean range.
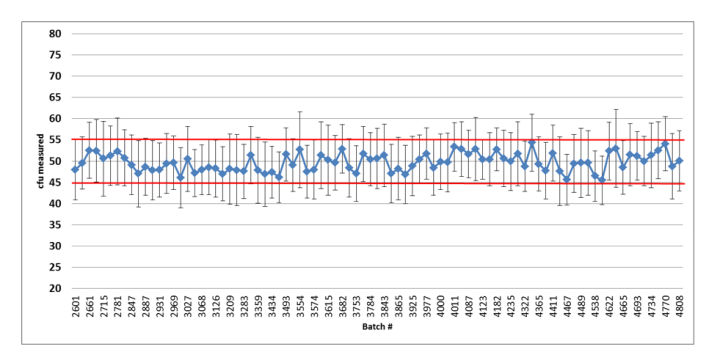
Figure 2: Summary graph of the quality control results of the released Bioball 550 batches of Bacillus subtilis: error bars represent standard deviation based on the repeated aliquots; Red lines indicate accepted mean range
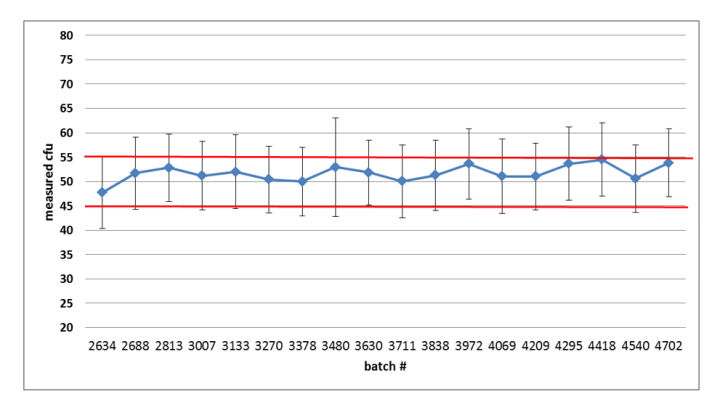
Figure 3: Summary graph of the quality control results of the released Bioball 550 batches of Burkholderia cepacia: error bars represent standard deviation based on the repeated aliquots; Red lines indicate accepted mean range
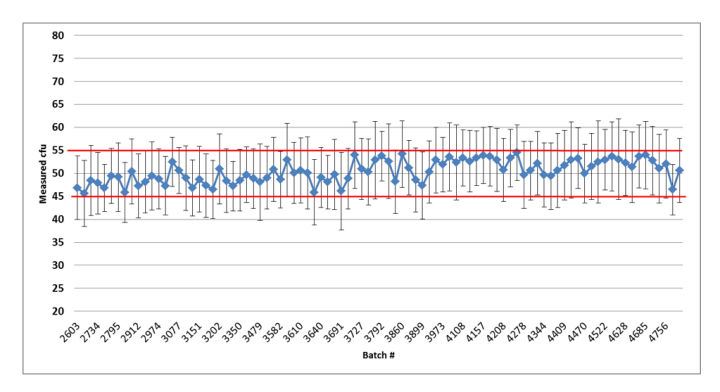
Figure 4: Summary graph of the quality control results of the released Bioball 550 batches of Candida albicans: error bars represent standard deviation based on the repeated aliquots; Red lines indicate accepted mean range; Red lines indicate accepted mean range
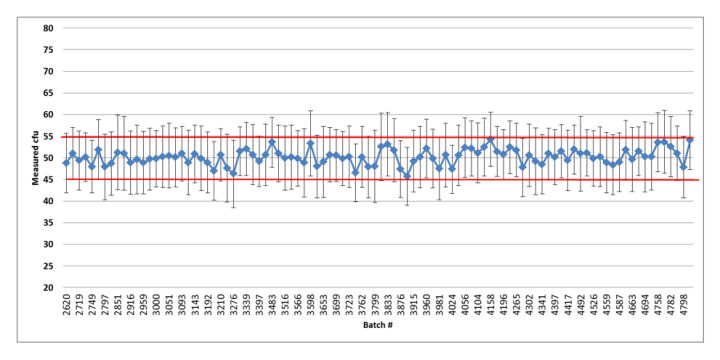
Figure 5: Summary graph of the quality control results of the released Bioball 550 batches of Pseudomonas aeruginosa: error bars represent standard deviation based on the repeated aliquots; Red lines indicate accepted mean range
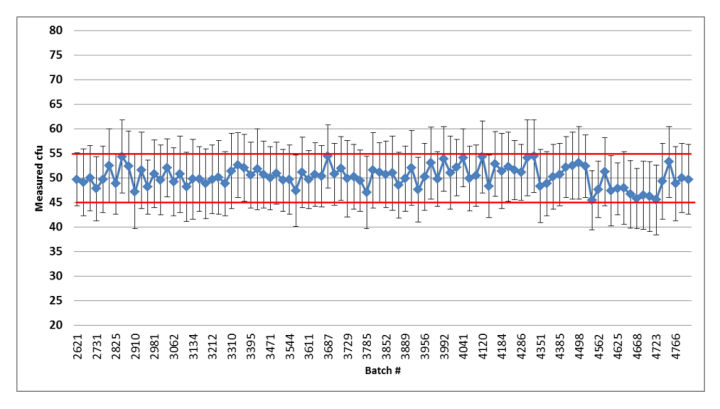
Figure 6: Summary graph of the quality control results of the released Bioball 550 batches of Staphylococcus aureus: error bars represent standard deviation based on the repeated aliquots; Red lines indicate accepted mean range
Results – Batch Stability (13 and 25 months)
As per internal procedure, a small selection of the released batches are tested for stability at mid and end of shelf life time. Table 2 shows a summary of this testing per species. It can be attested that all tested batches remained stable over 25 months (13 and 25 months average cfu within 10% of the original average measured cfu).
Table 2: Summary of long-term stability resuls per investigated species
| Organism |
Original CFU-Mean |
CFU 13 months - Mean | CFU 25 months - Mean | n |
| Aspergillus brasiliensis | 49.9 | 46.2 | 48.1 | 3 |
| Bacillus subtilis | 47.0 | 46.3 | 47.4 | 3 |
| Burkholderia cepacia | 51.2 | 49.3 | 49.3 | 9 |
| Candida albicans | 49.0 | 44.8 | 46.3 | 8 |
| Pseudomonas aeruginosa | 49.6 | 45.9 | 46.9 | 6 |
| Staphylococcus aureus | 50.8 | 50.3 | 50.6 | 4 |
Results - Moving range
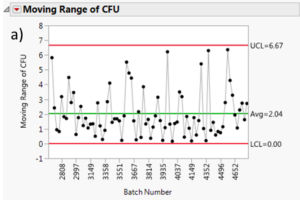
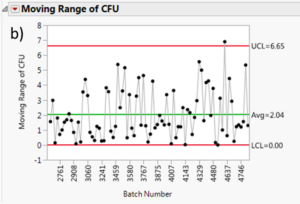
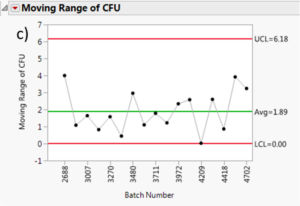
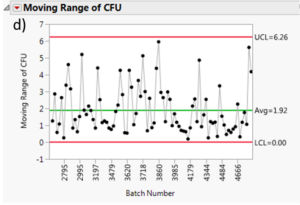
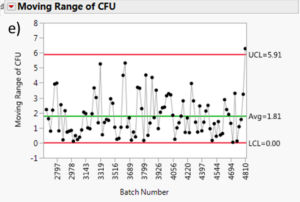
The moving range is a useful tool to monitor process variation. The moving range is defined as the difference between data point and its predecessor. In our case, it would be the difference in measured cfu mean from a batch and its predecessor. This measurement can help quality control personnel maintain an eye on short term bursts of variation and possible longer term trends. Figure 7 summarizes the moving range charts for each species. It can be assessed that the moving ranges are quite low for all the different Multishsot product presented here (average less than 3 cfu and maximum upper control limit (UCL) of 6.7 cfu).
 |
 |
 |
 |
 |
 |
Figure 7: Moving range charts for all investigated species: a) Aspergillus brasiliensis, b) Bacillus subtilis, c) Burkholderia cepacia, d) Candida albicans, e) Pseudomonas aeruginosa and f) Staphylococcus aureus
Conclusion
The data presented in this review reveals that all released batches from 2014 to 2018, across all commercialized organisms, are well within established specifications and show high level of reproducibility over time (average moving range of less than 3 cfu across all organisms). All real time stability results are also within specifications (13 and 25 months average cfu within 10% of the original average measured cfu). Based on the presented results, we could infer that, notwithstanding major changes in internal procedures or major deviations, the accumulated expertise regarding the 550s will continue to yield a reliably accurate and precise product.
 |
About the author: Brendan Tindall is the BIOBALL - Global Solutions Manager at bioMerieux. Brendan has almost 15 years of experience in various roles in Healthcare including Lifesciences, Medical devices and Pharmaceuticals. He has worked in local, regional and global roles. Brendan currently resides in Japan. Brendan holds a bachelor of science and a masters of management. |