Cherwell Laboratories, specialist suppliers of environmental monitoring and process validation solutions for the pharmaceutical and related industries, has launched a Redipor® prepared media distributor resource centre website. Developing this online knowledge hub supports Cherwell’s growing international distribution network within Europe and now Asia. This means that all Redipor microbiological media customers will receive quality support for their environmental monitoring, sterility testing, and process validation applications, whatever their location.
The new Redipor distributor resource centre securely houses lots of useful material, including prepared media product information, safety data sheets, guides, and updates which are readily accessible to the distributor at all times. This detailed information in turn enables them to help ensure the controlled environment monitoring efforts of Redipor users run as smoothly as possible. Further supporting in-house expertise, Cherwell invests time in developing a Redipor product champion at each of its distributors; training them in its processes and the complete range of Redipor culture media products, including features, benefits, and applications. This is all key to ensuring customers are advised on the right solutions for their needs.
“We are proud that the Redipor range has for many years been associated with quality products supported by an efficient, responsive, and truly flexible service for small and large users alike. We, therefore, take great care in selecting the right partners to work closely with us to supply our products internationally,” said Andy Whittard, Managing Director, Cherwell Laboratories. “Ensuring that all our distributors can also offer quality industry expertise and product support is crucial to us. This is because maintaining cleanrooms and other aseptic areas to the most stringent regulations, plus validating sterility testing processes, not only requires high quality prepared culture media but also quality in customer care.”
Cherwell’s network of expert distributors has been growing in Europe in recent years, now it has also carefully identified partners within Asia who understand the needs of the pharmaceutical and associated industries involved in aseptic processing. Cherwell has recently appointed two new distributors in the region, these are SPD Scientific in Singapore and Trans-Enor in Taiwan. These new distributors, along with ATS Korea in South Korea, are significantly raising the profile of Cherwell’s quality Redipor prepared microbiological media products in Asia. All of Cherwell’s distribution partners can be found on its website
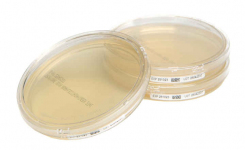
Redipor prepared culture media products are widely used in pharmaceutical, compounding, hospital pharmacy manufacturing units, and other related industries where cleanroom-based manufacturing is required. The Redipor range offers a vast selection of contact plates and petri dishes, plus liquid media in a variety of formats. Bespoke culture media options to meet specific needs are also available. Cherwell’s Redipor manufacturing facility is ISO 9001 registered, with all products undergoing stringent quality tests and QC certification. 100% inspection processes, gamma irradiation, and special packaging further deliver absolute confidence in product quality.
For more information please use the green “Request Information” button below.