APAS-AMR is the newest addition to Clever Culture Systems’ intelligent automation solutions for microbiology laboratories. Built using AI-driven image analysis, the APAS-AMR Analysis Module, deployed on the APAS Independence, offers a unique solution for antimicrobial susceptibility testing. APAS-AMR provides laboratories with rapid, high-throughput automated reading of disc diffusion plates processed in accordance with either CLSI or EUCAST methodologies. This is in addition to providing laboratories with the flexibility to read any panel of antimicrobials, allowing seamless integration with current practices.
Implementing the APAS-AMR Analysis Module within the laboratory is simple as it has been carefully designed to integrate within existing laboratory workflows and offers an affordable solution to address antimicrobial resistance testing needs. APAS-AMR improves accuracy and consistency of results by eliminating errors associated with subjective human interpretation and enhances record-keeping, using digital image capture for greater quality control. Additionally, the APAS Independence utilised with the APAS-AMR Analysis Module can be integrated with the Laboratory Information System (LIS) to deliver results directly. APAS-AMR delivers rapid, accurate, and repeatable results to clinicians, helping to decrease the time to optimise antimicrobial administration for patients. The reliability of APAS-AMR makes it a valuable tool in any antimicrobial stewardship program.
APAS-AMR features and benefits
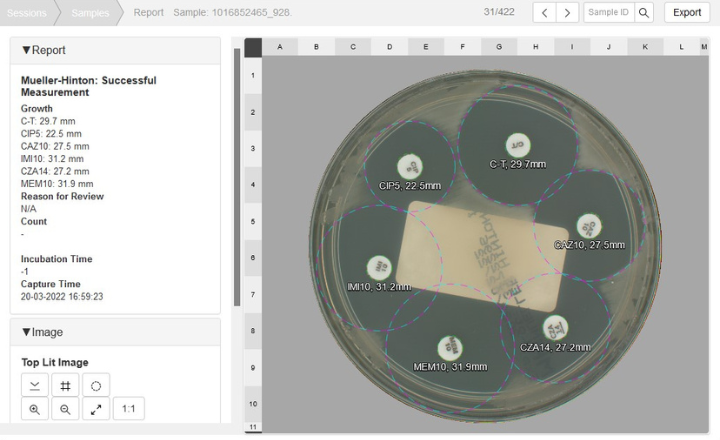
- Zone measurement - Accurate measurement of the zone of inhibition.
- Automatic disc interactions detection - Detection of D-zone and keyhole effects.
- Automatic disc recognition - Unique image-based antibiotic disc recognition means no need for preconfiguring disc panels.
- S.I.R. interpretation - Optional feature supporting CLSI and EUCAST breakpoints for S.I.R determination.
- Quality control features - Includes lawn quality check.
- Reporting - Sample reports are displayed via the APAS web interface, accessible from any connected workstation for easy review, with results transmitted to the LIS.
Click here to find out how APAS-AMR can enhance your laboratory’s antimicrobial stewardship program, or use the Request Information button to contact the supplier.