Updated: 21st Oct 2019
Key Points
- Breath and Stool tests are the most accurate non-invasive methods and only indicate active infections
- Serological testing can give false positives due to serological scars from previous infections
- Rapid non-invasive methods offer significant cost savings over invasive endoscopy
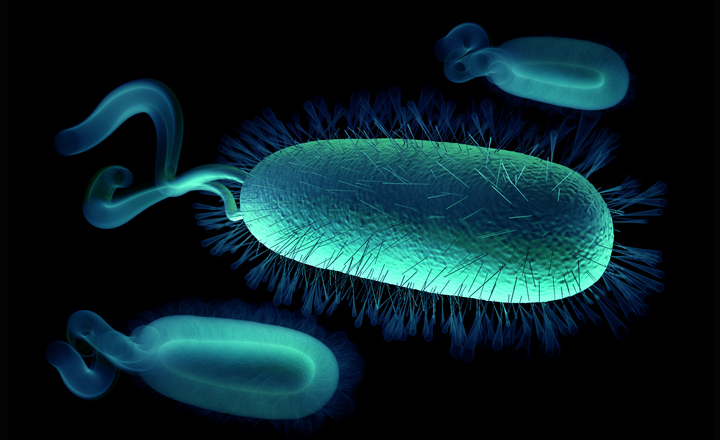
Helicobacter pylori is a spiral-shaped, Gram negative, microaerophilic bacterium commonly found in the gastric mucosa lining the upper gastrointestinal tract (stomach and duodenum) of humans and animals.Transmission of infection is thought to be via the faecal-oral, or oral-oral route, but the exact mechanisms remain uncertain. However, infection is certainly contagious, and about two thirds of the global population is estimated to be carrying H. pylori, with infection being significantly more common in developing countries. The discovery that H. pylori infection is the major cause of gastritis and gastric and duodenal ulcers has revolutionised the treatment of these conditions. Antimicrobial therapy has largely replaced the previous practice of long-term acid suppression medication, which treated the symptoms, but not the underlying infection.
Many people infected with H. pylori suffer no related symptoms, but infection can cause chronic gastritis in adults and children, both gastric and duodenal ulcers and an increased risk of gastric cancer. Infection can result in an immune reaction leading to a localised inflammation of the lining of the stomach and duodenum. Ammonia and other toxic substances produced by the bacteria may also damage epithelial cells and contribute to the inflammation. Inflammation may also increase stomach acid production and damage the gastric mucus layer. This increased exposure to stomach acids can cause ulceration and possibly stomach cancer in some cases.
Detection and diagnosis:Testing for H. pylori infection has become a very important part of the diagnostic process for gastric and duodenal inflammatory disease, since the presence or absence of infection determines the type of treatment to be applied. Testing is also a useful means of monitoring the effectiveness of courses of antimicrobial treatment. A number of different diagnostic test methods, both invasive and non-invasive, are available.
Non-invasive methods
Several techniques for non-invasive diagnostic tests for H. pylori have been developed. These tests have been introduced for low-risk patients and are in widespread use:
Stool tests Recently, new tests for H. pylori infection have been developed that rely on the detection of specific antigens in the stools of infected individuals. These are usually EIA-, or amplified EIA-based tests and several are available in a lateral flow format for use at the point-of-care. Stool antigen testing has been found to be reliable, inexpensive and easy to use. It also has the advantage of indicating only active infection. This means that it can be used to monitor the eradication of infection by antimicrobial treatment and can also detect repeat infections.This test is highly sensitive and has outstanding specificy. It requires no special set-up, patient fasting, dietary restrictions or avoidance of other medication.It is cleared for use in patients of all ages.
Breath test The breath test utilises the ability of H. pylori to produce large quantities of urease as a diagnostic characteristic. The patient is required to drink a solution of urea labelled with 13C, or 14C isotopes. If an H. pylori infection is present, the urea will be metabolised by the bacteria, producing ammonia and isotope-labelled carbon dioxide, which can be detected in the patient’s exhaled breath by radioactive counting or mass spectrometry. A breath test based on 13C-labelled urea and using infrared spectrophotometry to measure the ratio of 13C to 12C in a breath sample has been developed into a commercially available product for use at the point-of-care. Breath testing has been found to have a high sensitivity and specificity (94-98%) and can be applied at moderate cost. It is also suitable for monitoring the effectiveness of treatment as it is specific to active infections and can be used to confirm eradication.
Serological tests This is the most common method used to test for H.pylori. However, its accuracy is undermined by a patient's "serological scars" i.e antibodies remaining in the patient's blood from a previous infection. A serological test can show positive for H.pylori even after the patient has recovered from that infection. Due to the inevitable misdiagnosis, the use of serological testing for detection of H.pylori is being phased out in the United States.
People infected with H. pylori generally have specific IgG and IgA antibodies circulating in their blood and these can be detected by serological tests. Most of the commercial tests available are based on IgG detection, which is considered slightly more sensitive and specific. Antibody detection is usually by laboratory-based qualitative enzyme immunoassay (EIA) methods presented in a microplate assay format, although automated immunoassay systems have also been developed. Rapid tests that can be applied at the point-of-care are also available, either based on immunochromatographic technology (lateral flow tests) or on latex agglutination. Quantitative tests are considered to be of limited value.
it is less time consuming and much less costly than invasive methods and means considerably less discomfort for the patient. However, as aforementioned , serological testing cannot be used to monitor the progress of antimicrobial therapy, since patients whose infection has been eradicated may continue to carry serum antibodies specific to H. pylori for several months after successful treatment.
Invasive methods
The collection of biopsy specimens from inflamed or ulcerated regions of the stomach and duodenum during invasive endoscopy is considered to be the reference method for diagnosing H. pylori infection. The biopsy material can be examined using one, or more, of three different test methods.
- Histological examination – staining and examination of the tissue samples allows both evaluation of cell damage and the detection of H. pylori cells in situ. Histological identification is currently regarded as the ‘gold standard’ of diagnostic tests.
- Urease test – a colorimetric test for the presence of urease enzyme activity (indicative of H. pylori infection) in the biopsy sample. This method can be used to give a rapid indication of infection at the time of the biopsy.
- Culture and isolation of H. pylori – the tissue sample may be homogenised, or inoculated directly onto selective agar media. Typically, cultures are incubated for at least 3-5 days at 35oC under microaerophilic conditions. Isolates can be confirmed as H. pylori by Gram staining, urease reaction and other traditional identification tests. PCR-based methods have also been developed, but are not yet widely available. Culture and isolation is necessary if antimicrobial susceptibility testing is required.
Invasive testing is considered to be highly specific – particularly histological examination – but its sensitivity is partly dependent on the accuracy of the biopsy procedure, since sampling tissue from areas where only low numbers of bacteria are present may give false-negative results. Endoscopy is also an expensive and demanding procedure requiring highly trained, skilled staff and is quite uncomfortable for the patient.
Both histological examination and culture of biopsy samples are reliable methods, but they are time consuming and require specialised laboratory facilities with highly trained staff. Urease testing is much more rapid and less costly, but still requires an invasive procedure to obtain the samples.
History The presence of spiral shaped bacteria in the stomach mucosa of people with gastric disease was first noted over 100 years ago, but the observation was not investigated thoroughly until the late 1970s, when an Australian pathologist, John Warren, noted that spiral-shaped bacteria could often be seen associated with inflamed gastric tissue. Together with physician Barry Marshall, Warren conducted further research on the organism and finally succeeded in isolating and culturing it in 1982.
Warren and Marshall put forward the hypothesis that the bacterium – called Campylobacter pylori until 1989 when genetic analysis revealed that it should be placed in a new genus Helicobacter – was, in fact, the major cause of human gastritis and gastric ulcers. At the time, the accepted view was that factors such as stress, excess acid and spicy foods were the cause of these conditions and the medical establishment received the hypothesis with some scepticism. Famously, Marshall demonstrated that Koch’s postulates could be met for Helicobacter pylori as a cause of gastritis by the simple expedient of drinking a culture of the bacteria. Sure enough, he became ill with nausea and vomiting after several days and was also able to show that the infection could be cured by antibiotic treatment. By the late 1980s, other research groups had been able to confirm Warren and Marshall’s hypothesis and the role of Helicobacter pylori in gastric illness became generally accepted.
Pathogenicity One of the reasons why physicians were reluctant to accept the ideas of Warren and Marshall was the harsh acid environment of the stomach, which had always been thought of as virtually sterile. How could a bacterium survive and infect the epithelial lining of the stomach in such conditions? It was eventually discovered that H. pylori has some useful adaptations that allow it to do just that. Firstly, it is motile and able to invade and colonise the thick mucus layer protecting the stomach wall from acid damage. From there it can reach the underlying gastric epithelium, where the bacteria then bind to the epithelial cells. It is also able to manipulate its environment using a urease enzyme to convert urea in the stomach to ammonia, which raises the pH around the cell and helps to protect it from stomach acid. The presence of urease is an important diagnostic characteristic for H. pylori.
It is now known to have important implications for human health, but its significance is a comparatively recent discovery.
Get the latest updates in Rapid Microbiological Test Methods sent to your email? Subscribe to the free rapidmicrobiology eNewsletter