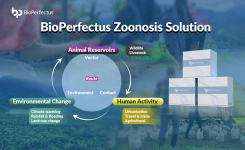
Monkeypox is a viral zoonosis with symptoms very similar to those seen in the past in smallpox patients. lt is caused by the monkeypox virus which belongs to the orthopoxvirus genus of the Poxviridae family. Monkeypox presents with fever, an extensive characteristic rash and usually swollen lymph nodes.The incubation period of monkeypox is usually from 6 to 13 days but can range from 5 to 21 days. WHO recommends that detection of viral DNA by polymerase chain reaction (PCR) is the preferred laboratory test for monkeypox.
Key Points:
- Innovative 3-in-1 tube PCR
Innovative single-tube PCR, with reaction solution, primers and PCR enzymes in one tube,minimize time of sample adding - Various Specimen
Oropharyngeal swab, vesicular or pustular lesion fluid, diseased tissue - High Sensitivity
Reliable detection of Monkeypox virus, the limit of detection can be up to 200 copies/mL - More Reliable
Based on Polymerase Chain Reaction(PCR) method, the gold standard for diagnosing monkeypox infection recommended by WHO - More applicable
Widely applicable in instruments with FAM,CY5 fluorescence channels