This year's Annual PDA Global Conference on Pharmaceutical Microbiology in Rockville, MD, Oct 21 - Oct 23, 2019, continues the ongoing tradition of addressing the opportunities and challenges most relevant to the future of microbiology in today’s global market. Whether you are attending the conference or just want to keep up to date with the latest in products for the pharma microbiology laboratory, you can browse the latest updates that will be on the exhibition stands.
Explore Charles River Microbial Solutions at PDA Microbiology 2019
|
Find out why you should visit Charles River Booth #209 at this year’s PDA Global Conference on Pharmaceutical Microbiology. |
Let’s Explore Endotoxin Testing Solutions at PDA Microbiology 2019
|
Speak with Lonza Experts about your endotoxin testing challenges at Booth #310. Let’s solve them together. Additionally, see our poster presentation “Establishing the Monocyte Activation Test (MAT) in a New Lab - Assay qualification and product-specific validation. |
Time-tested Quality with MilliporeSigma. What’s new at Booth 313?
|
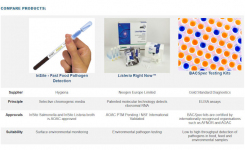
Come visit MilliporeSigma at PDA Micro Booth #313 and be the first to see the latest improvements in your daily testing workflows. |
Ensuring Data Integrity Using PDA TR80 and ALCOA/ALCOA+
|
Rapid Micro Biosystems breaks down how to successfully use the TR80 framework and ALCOA/ALCOA+ that helps to support the FDA’s agency stated efforts to improve drug quality through vigilant oversight of data integrity and good manufacturing practice. |
Still Using Paper to Manage Your QC Data?
|
Replace paper with productivity - the MODA™ Platform bridges the gap that currently exists between manufacturing and QC to provide a single batch record with an intuitive review and approval interface. The MODA™ Platform has GMP compliance and tracking at its core. |
One-hour Mycoplasma Testing by Anyone, Anywhere, Anytime.
|
Do you think Fast, Easy and Accurate Mycoplasma testing is a fantasy? It's not. bioMérieux has simplified complexity with the BIOFIRE® Mycoplasma “Lab in a Pouch”. |
How to Ensure your Data Integrity with ScanStation, Real-time Bacterial Monitoring
|
ScanStation, real-time bacterial monitoring unit, detects and counts colonies as soon as they appear at the beginning of the incubation. It provides anticipated results for quality control labs with full data integrity 21 CFR part 11. |
Faster, Easier, Better Contamination Monitoring with Rapid Sample Prep .
|
The InnovaPrep CP Select is a rapid and automated concentration tool for contamination monitoring. It exponentially improves sensitivity for nearly any analytical method so you can get same-shift results while improving your LOD. |
|
Meet USP requirements with test-ready microbial controls for Growth Promotion Testing and Environmental Monitoring controls. |