Platform Puts COVID-...
High-quality Solutio...
29th September 2020 Product update: rapidmicrobiology staff writer
Pasteur Institute Start Clinical Trial on Point-of-Care COVID-19 Test
C4Diagnostics, Bertin Technologies, and Enalees have announced the launch of their clinical trial of BEC-SARS-CoV-2, a point-of-care test to detect SARS-CoV-2 in patients, which was developed jointly with Pasteur Institute.
The BEC-SARS-CoV-2 clinical diagnostic test has been submitted to the authorities per French regulations, the detection process has been validated and a clinical trial has been launched by the consortium to obtain CE-IVD marking by the end of September.
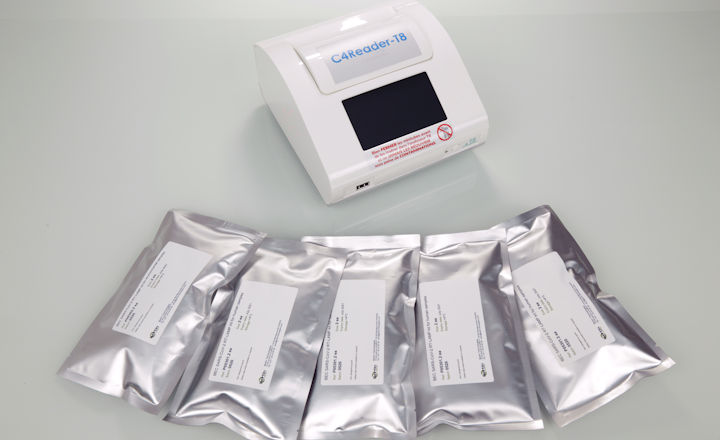
In the meantime, the test is available as RUO. This sensitive, rapid, and portable diagnostic test rely on two innovative technologies for the extraction of viral RNA and its rapid detection using RT-LAMP.
Due to its portable format, it can detect SARS-CoV-2 in humans at point-of-care within 25 minutes (from collection to result) from a nasal or nasopharyngeal swab. It has an endogenous control and targets two different genes (in line with the World Health Organization and the French Health Authority (HAS) recommendations).
Laurent Thiery, CEO and Co-founder of Enalees, states: "The BEC-SARS-CoV-2 for humans test kit is based on the Enalees test concept that we have been using for two years to detect and curtail the transmission of respiratory diseases, on-site, in domestic animals such as horses.”
He continued: “In particular, our rapid viral RNA extraction technique validated previously on test kits for animal diseases is at the core of this new test kit. In addition, this test also uses a rapid molecular diagnostic technology based on RT-LAMP, used in all Enalees test kits and during previous health crises such as Ebola or Zika by Pasteur Institute.”
The results C4Diagnostics has obtained are currently being tested in a prospective clinical trial led by Pasteur Institute in several clinical centres.
This clinical data will confirm the sensitivity and specificity of the diagnostic test and C4Diagnostics are expecting to have BEC-SARS-CoV-2 for humans CE-IVD marked by the end of September.
There is also a BEC-SARS-CoV-2 kit to detect the virus in the environment, which was developed and marketed in May.
It is used in conjunction with Bertin's Coriolis air sampler and provides a comprehensive solution for analysing contamination in the environment.
Combined, the two BEC-SARS-CoV-2 tests allow the detection of COVID-19 in a contaminated person's social group and their environment and developed to stop all new outbreaks of SARS-CoV-2 in a controlled and localised way.
BEC-SARS-CoV-2:
This field diagnostic test kit uses RT-LAMP technology, which has an equivalent sensitivity to RT-PCR for the detection of SARS-CoV-2.
RT-LAMP is an amplification method that, unlike traditional RT-PCR, does not require temperature cycles for the amplification of RNA.
The advantages of this technique are highlighted in its speed, ease of use, and robustness. Amongst the various existing isothermal amplification methods, LAMP has gained particular popularity over the last few years. As an example, the WHO recommended its use for the diagnosis of tuberculosis.
In terms of robustness and performance, this test satisfies the requirements of the French National Health Authority (HAS). It has two viral target genes for better sensitivity and specificity, and an endogenous positive control to ensure both the quality of the sampling technique and the correct functioning of the test and equipment, thus limiting the number of false negatives.
Tags:
Date Published: 29th September 2020
Source article link: View
Note: This content has been edited by a rapidmicrobiology staff writer for style and content.
Platform Puts COVID-19 Vaccine Developers
High-quality Solutions for High-quality Beverages