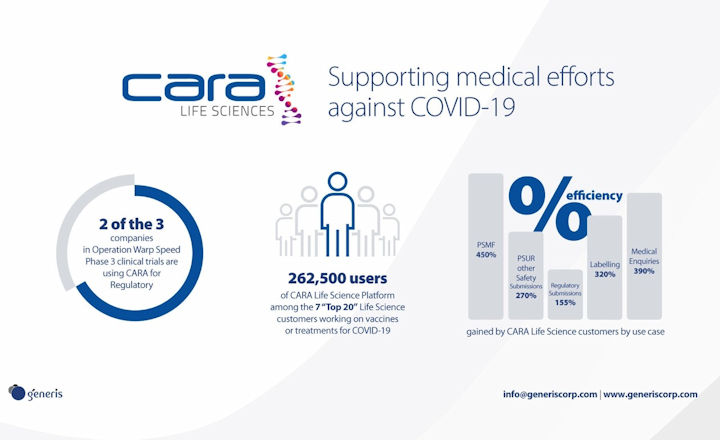
Generis is proud to announce that two out of the three leading COVID-19 vaccine initiatives in Operation Warp Speed are using the CARA Life Science Platform for Regulatory.
The US-driven international initiative to support the rapid development, manufacturing, and distribution of COVID-19 vaccines is progressing with many promising efforts to move towards Phase 3 clinical trials. The CARA Life Sciences Platform, provided by Generis, is helping these Life Science companies to greatly reduce their development and submission cycles while focusing on oversight and compliance with stringent health regulations.
The CARA Life Sciences Platform manages regulated business processes, managing content and data across the core functional areas of Life Science companies. Adoption of this platform has driven process efficiency improvements of up to 450%*, greatly reducing time-to-market for regulated products.
*measured based on average time generating a PSMF annex in legacy solutions or processes that have been replaced by CARA
Note: This content has been edited by a rapidmicrobiology staff writer for style and content.