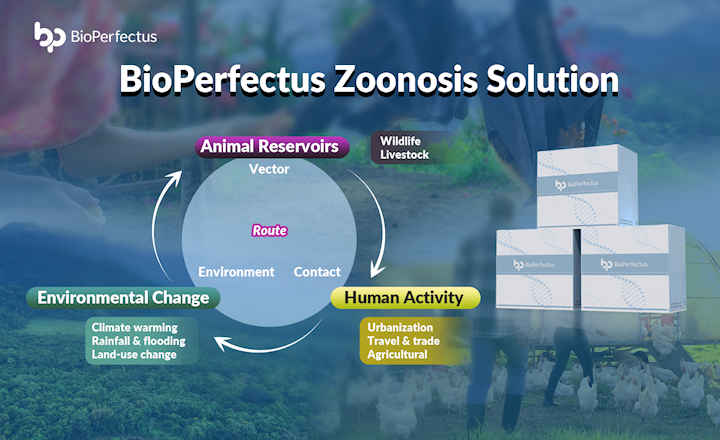
BD (Becton, Dickinson and Company) working together with CerTest Biotec have made their newly developed PCR assay for monkeypox virus commercially available worldwide, including in the United States, for research use only (RUO) by laboratories.
The BD MAX™ System family is a fully integrated, automated platform that does nucleic acid extraction and real-time PCR. In less than three hours, it can give results for up to 24 samples from different syndromes.
The CerTest VIASURE Monkeypox molecular RUO assay was made by the teams with the help of the BD MAX™ System open architecture reagent suite. Based on what the FDA said on September 7, BD is working to get an emergency used authorization (EUA) for a clinical test for the monkeypox virus as soon as possible.
The BD MAX™ System offers a wide range of tests for infections related to health care, respiratory infections, sexually transmitted infections, GI infections, and women's health.