Dutch biotechnology company Micreos have secured €30 million in funding to accelerate the development of its endolysin technology, set to replace antibiotics. Proceeds are earmarked for the clinical development program of its endolysin XZ.700 and the USA launch of its breakthrough OTC Gladskin product for eczema.
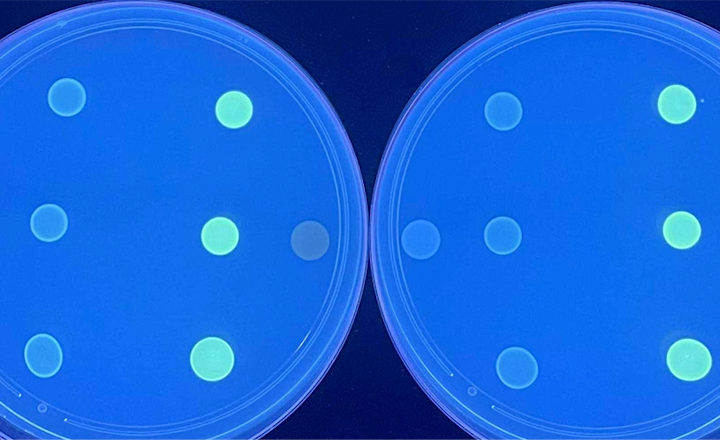
Endolysins have three distinct features:
- ability to target only unwanted bacteria while preserving the microbiome
- ability to kill antibiotic resistant strains of bacteria
- emergence of resistance against endolysins is not expected.
Endolysin technology opens a new therapeutic window for treatment of chronic inflammatory conditions, such as atopic dermatitis and persistent wound infections, including those caused by MRSA.
Micreos' Staphefekt™ SA.100, is the world's first endolysin approved for human use. It selectively targets Staphylococcus aureus - a major trigger of eczema and other inflammatory skin conditions.
Video: How Staphefekt™ works in eczema: https://youtu.be/qxg2kxfFw6Q
"Since its introduction, Gladskin has helped over 100.000 people in Europe with eczema, acne, and rosacea, many of them reporting a life-changing impact,'' remarked Skyler Stein, Head of Gladskin USA. "Gladskin is pioneering the use of endolysins to rebalance the skin microbiome and improve skin health." Chosen as Europe's Most Impactful Innovation 2018, Gladskin is now preparing its launch in the United States.
The €30 million funding includes a non-dilutive €5.4 million Innovation Credit granted by the Dutch Ministry of Economic Affairs. Micreos has conducted several studies with Gladskin, including trials on eczema, acne and rosacea. Studies in orphan indications with a high unmet need, like primary immune deficiencies and Netherton disease are ongoing.