In this exclusive interview with rapidmicrobiology.com, Dr. Andrew Pridmore, Head of Microbiology at Don Whitley Scientific, discusses the benefits of using a spiral plater, what to look for in a system and how the new WASP Touch from Don Whitley Scientific (DWS) can help ensure your laboratory has consistent trackable, plating results.
Don Whitley Scientific introduced their first Spiral plater in 1978, why are they still relevant to today’s modern microbiology lab? Much of the testing done back in 1978 when we first introduced spiral plating to the UK, is still relevant today. However, increased quality control measures have led to laboratories looking more to automation to help improve reproducibility and accuracy. Spiral plating offers a real reduction in the cost-per-test and standardises counting methods. Serial dilutions, for many users, can be eliminated, which in turn saves time, the cost of consumables and space in the incubator.
One of the key issues facing labs is calibration, validation and traceability, how can a spiral plater help with this? Spiral plating techniques are recognised in the ISO 4833-2 standard. This is a general recognition of the use of spiral plating techniques in food microbiology and provides standardised procedures for their use.
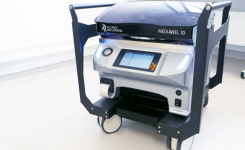
Many spiral plating processes cannot be calibrated but with the WASP Touch, users can calibrate the delivery of the sample. There is also the option to include a bar code scanner. Traceability is facilitated with the inclusion of the Data Handling package for WASP Touch. This option records date, time, user, plate id, volume, date and at which volumes, date of the last start and finish position check, and date the last dye plate was performed. It also provides multi-level PIN-code protection. Data can be downloaded via USB in a format that can be loaded directly into Excel. An Ethernet connection for data transfer/service support is also included.
When choosing a new spiral plater what features should a buyer be looking for? An automated spiral plater is ideal for those who regularly need to plate samples with a microbial content greater than 1,000 CFU/ml. Buyers should look for a plater that is flexible enough to process their particular samples, in terms of quantity and type of sample. For example, WASP Touch offers an Extended Volume Package to enable a wider range of volumes on a plate. Buyers should also consider the level of after-sales service, maintenance and support offered by the manufacturer. If you are opting for an automated method to speed up your processes, the last thing you want is for there to be a problem that causes an unacceptable level of downtime.
If a lab is getting an in-house demo for a spiral plater do you have any recommendations for how to test it? At Don Whitley, we feel that any test should be as realistic as possible so would always look to perform a demo or trial with the actual samples the laboratory uses. After working through a demonstration with the customer, we would ideally arrange for them to have a spiral plater for an extended trial. This helps them to make a more informed purchase decision.
DWS have recently introduced a next generation spiral plater, what issues does this model address? The new WASP Touch spiral plater simply modernises the process of spiral plating. The user interface is now a touchscreen with easy-to-use icons. Other features such as FlowSense ensure that the sample is correctly processed, alerting the user to any issues in time for corrections to be made; and AIMS, the intuitive software that guides users through the set-up routine.
If a lab wanted to introduce the new DWS spiral plater, what validation steps would be needed (is that the same if they were introducing spiral plating for the first time or upgrading to the new DWS system)? WASP Touch, and spiral platers in general, require minimal validation because spiral plating methodology has already been validated and published in the international standard ISO 4833-2:2013. The ISO standard includes methods for calibration and validation of the sample volumes deposited onto an agar plate by a spiral plater.
Click here for more information on the Whitley WASP Touch

Dr Andrew Pridmore is Head of Microbiology at Don Whitley Scientific. Dr. Pridmore has many years’ experience of running the GLP-compliant, contract microbiology laboratories at DWS where spiral plating is routinely used.