Current qPCR-based methods for screening STEC have the common challenge in differentiating between samples where a single organism contains both stx and eae (true positive, linked virulence) and samples in which stx and eae reside in different organisms (false positive, unlinked virulence). Bio Rad offers a Droplet Digital PCR solution, the dd-Check STEC Solution, that demonstrates the capacity in virulence linkage analysis by partitioning intact cells into droplets where cell lysis and PCR amplification occur, enhancing the test accuracy by reducing false-positive reactions.
To evaluate the ability of ddPCR technology in detecting and distinguishing E. coli cells with linked and unlinked virulence compared to qPCR technology, a collaborative study between Food Safety Net Services and Bio Rad was performed. A set of three matrices, beef trim, ground beef and MicroTally (n=30), were inoculated at < 5 CFU per 375 g sample. For each sample type, 15 samples were inoculated with one regulated STEC strain with linked virulence genes (stx and eae in the same cell), and 15 samples were inoculated with a cocktail of two regulated serotypes with unlinked virulence genes (one with stx only and one with eae only).
All samples were processed following two timelines for evaluation of the ddPCR technology as a primary screening assay compared to qPCR and a confirmatory assay, illustrated in Fig. 1. Following the timeline for using ddPCR as primary screening, samples were processed for detection of stx and eae via qPCR (iQ-Check STEC VirX Kit, Bio Rad) and ddPCR technologies (dd-Check STEC Solution, Bio Rad) after a 16 hr enrichment. Following the timeline for using ddPCR as a confirmatory assay, samples were processed for primary screening using qPCR after an 8 hr enrichment, followed by confirmation via ddPCR with or without regrowth according to the criteria demonstrated in Fig 1.
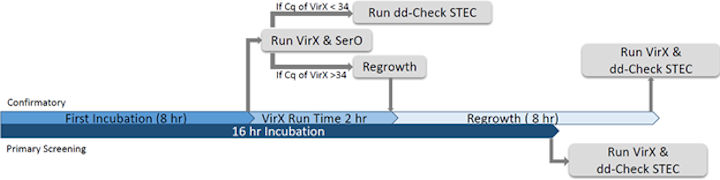
Figure 1. Sample processing workflow timeline.
Cultural confirmation of STEC in all samples was performed following the USDA MLG 5C.01 protocol. The results obtained by qPCR and ddPCR assays were compared against the reference method for accuracy in screening and distinguishing STEC with linked and unlinked virulence genes.
Results demonstrated that the ddPCR technology was able to individually identify the presence of stx and eae and verify the co-existence of these two virulence genes via linkage analysis (Fig 2).
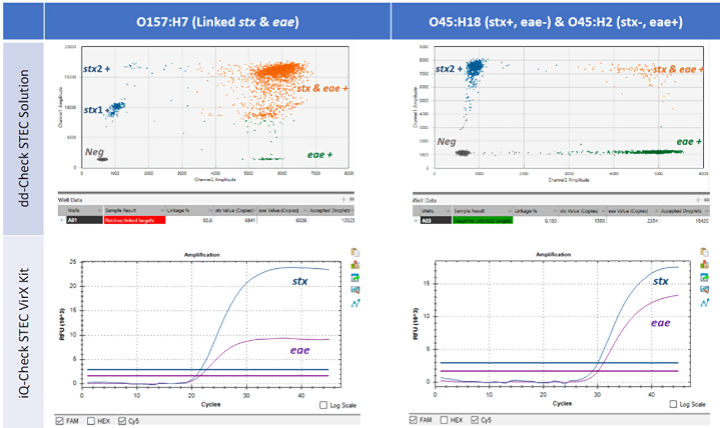
Figure 2: Results detecting linked and unlinked virulence genes.
The ability of ddPCR technology as a primary screening assay to identify the co-existence of stx and eae in a single bacterium enhances the accuracy of STEC testing by reducing number of false-positive reactions. In this study, ddPCR technology also proved useful for culture-independent confirmation following primary screening by qPCR assays.
To further explore the possibility of using ddPCR as a culture independent confirmation method, a second study that evaluated ddPCR for confirming STEC in routine beef samples following positive results from primary screening by qPCR was conducted. In this study, Bio Rad collaborated with Food Safety Net Services and several beef production facilities from four different states (Texas, Kansas, California, and Colorado) to collect a set of 100 routine beef microbiological samples (consisting of both MicroTally manual sampling devices and beef trim) that were presumptive positive for E. coli O157:H7 via qPCR-based primary screening tests over a period of six months. After presumptive positive results were obtained with qPCR assays, samples were sent to the Lab+ Department at Food Safety Net Services (San Antonio, TX) and were processed for culture-independent confirmation using the ddPCR technology (dd-Check STEC Solution, Bio Rad) and cultural detection and confirmation following the USDA MLG 5C.01.
Results for the confirmation of linked virulence profiles (i.e. organisms containing both stx and eae) showed that the ddPCR assay and the USDA MLG 5C.01 identified the presence of linked targets in 97 and 95 samples, respectively, indicating that ddPCR and the MLG reached a high level of agreement (98%) in confirmation of linked virulence profiles. Two samples were positive for linked virulence (presence of stx and eae in a single cell) via ddPCR and produced colonies that were positive for stx and eae, but the serotype of the colonies obtained was not able to be identified. In addition, for three samples, ddPCR provided an unlinked stx and eae profile, which matched the confirmation results produced with MLG 5C.01. With a high level of agreement in STEC confirmation between ddPCR technology and the USDA MLG 5C.01 method, ddPCR technology proved useful for culture-independent confirmation following primary screening by qPCR assays.
The ability of ddPCR technology to be used as a rapid confirmation assay to identify the co-existence of stx and eae in a single bacterium enhances the accuracy of STEC testing by narrowing positive results to only those that have linked stx and eae virulence markers. Additionally, ddPCR testing can be completed in a matter of hours as opposed to days with current cultural confirmation techniques.
To learn more visit or ask for details using the green "Request Information" button below.