Cherwell Laboratories, manufacturer of Redipor® prepared microbiological media, has completed an expansion programme at its Bicester-based facility. Initiated in response to a significant increase in demand, Cherwell has doubled the size of its cleanroom manufacturing suite and recruited additional staff to support the higher production capacity. Cherwell is renowned for offering a flexible supply of quality prepared media products, and the expanded facility ensures increasing demand in the UK and mainland Europe can be accommodated.
Cherwell’s continuing growth is partly fuelled by the development of export sales - up 55% in the past two years - and the appointment of further European distributors for the Redipor range. Already supported in Italy, Ireland, Poland and Slovenia, Cherwell has recently signed distributor agreements with AcefeSA in Spain and Dorte Egelund in Denmark. The Company has carefully selected partners with the capability to deliver the same quality of service enjoyed by its UK customers.
The Company has increased its UK market share through a dedication to customer service and the ability to deliver bespoke prepared media solutions. Many customers are switching to the convenience of Redipor pre-prepared media rather than making their own onsite. Increased pressure by regulators for more environmental monitoring has also led to overall growth in the prepared media market. Notably, demand for Redipor Barrier pack products, for use in isolators where Vaporised Hydrogen Peroxide (VHP) is present, has grown, reflecting the increased use of VHP technology for sterilisation procedures.
Andy Whittard, Managing Director, Cherwell Laboratories commented, “This investment in facilities and capabilities is a clear indication of our commitment to developing our position as a leading supplier of prepared media. Recent market research shows our customers value not just the quality of our products, but increasingly our service and support. Most importantly, we are consistently delivering what our customers want.”
He added, “By developing our export markets through carefully selected partners, we aim to continue growing our UK-based production in a controlled manner, keeping excellent service and the customer at the core of what we do.”
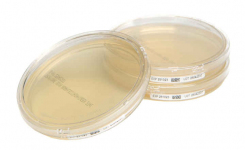
With over 40 years’ experience, Cherwell has developed a fully integrated range of products for environmental monitoring, product sterility testing, operator validation and process validation. Cherwell’s range of Redipor high-quality prepared media products includes petri dishes and contact plates, plus bottled media, broth bags and ampoules.
Redipor prepared media is designed to meet customer requirements for both industry standard products and those with a unique formulation or presentation. All Redipor products are subjected to a full array of QC tests, including comprehensive growth tests, with detailed QC certification supplied for every batch. The flexible production methods and managed stock levels, all supported by the new manufacturing facility and investment in additional production equipment, allow Cherwell to deliver cost-effective, in-time solutions for both large and small customers alike.
For more information about Cherwell Laboratories, please visit www.cherwell-labs.co.uk or follow @CherwellLabs on Twitter.