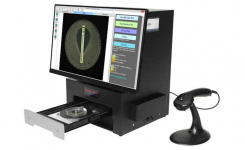
BIOMIC V3 is an open system utilizing digital imaging to automate the reading and CLSI and EUCAST interpretation of clinical microbiology tests and QC from various manufacturers.
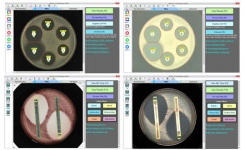
BIOMIC V3 systems are customized with optional modules: Disk Diffusion, 96-Well Microtiter, MIC Strip, Organism ID, Colony Count, and Urine Screen.
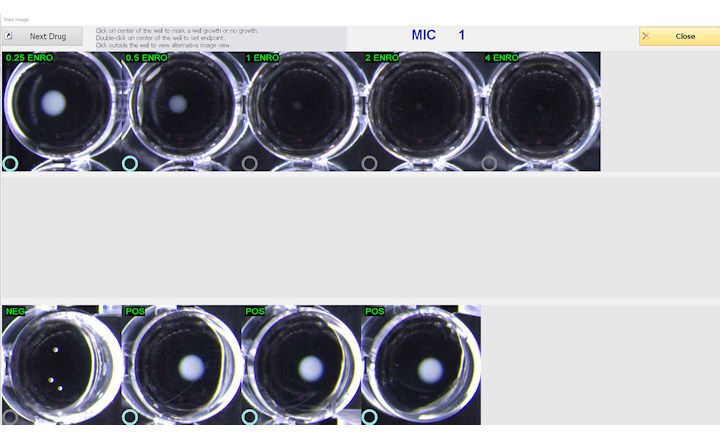
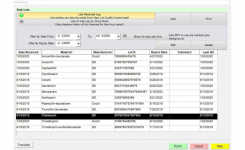
BIOMIC V3 provides a digital record of test results and high-resolution images. An LIS/LIMS interface combined with bar code reading and touch-screen entry on a 24-inch monitor offers labs an optimal setup to standardize, record and report test results.
Current CLSI guidelines updated in BIOMIC V3:
- M100, 31st edition: Performance Standards for Antimicrobial Susceptibility Testing
- VET01S, 5th edition: Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals
- VET04, 3rd edition: Performance Standards for Antimicrobial Susceptibility Testing of Bacteria Isolated from Aquatic Animals
- M60, 2nd edition: Performance Standards for Antifungal Susceptibility Testing of Yeasts
- M61, 2nd edition: Performance Standards for Antifungal Susceptibility Testing of Filamentous Fungi
Current EUCAST guidelines updated in BIOMIC V3:
- Breakpoint Tables for Interpretation of MICs and Zone Diameters Version 11.0
- Zone diameter breakpoints for rapid antimicrobial susceptibility testing (RAST) directly from blood culture bottles Version 3.0
- Routine and extended internal quality control for MIC determination and disk diffusion as recommended by EUCAST Version 11.0
BIOMIC V3 is designed and manufactured by Giles Scientific in Santa Barbara, California, USA. Please contact Giles Scientific for further information or click here to visit their website.