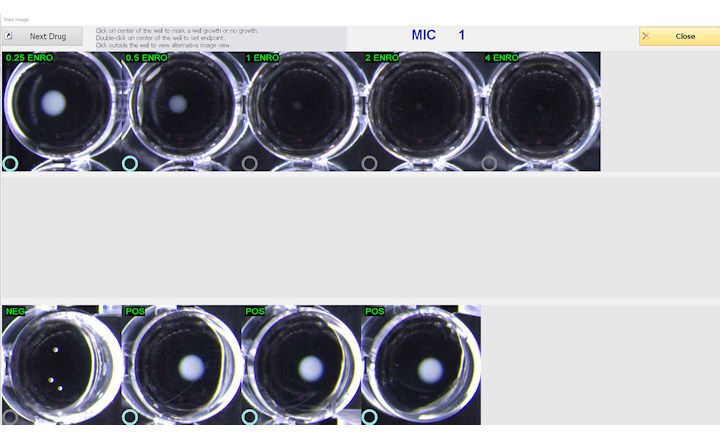
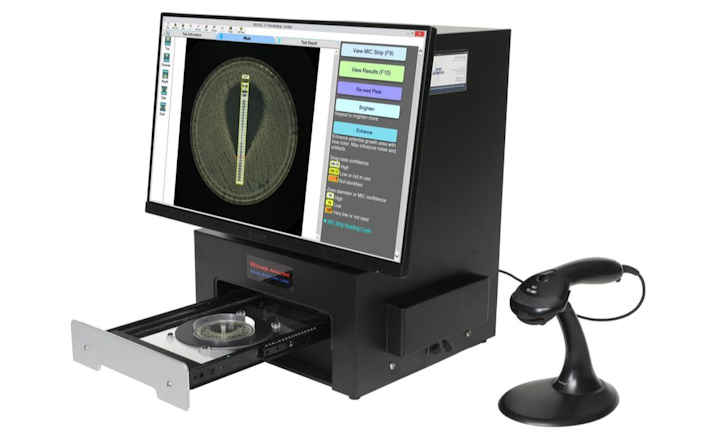
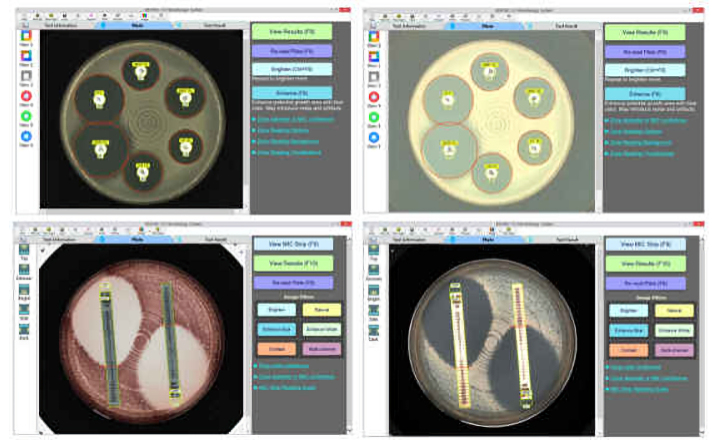
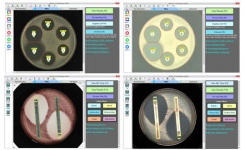
BIOMIC V3 is a digital imaging system for automating the reading and interpretation of AST and ID tests from various manufacturers.
The following guidelines are now updated in the 2019 BIOMIC V3 software:
- CLSI M100, 29th edition: Performance Standards for Antimicrobial Susceptibility Testing
- CLSI VET01 5th edition & VET08 4th edition: Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals
- EUCAST Breakpoint Tables for Interpretation of MICs and Zone Diameters v 9.0
- EUCAST Routine & Extended Internal QC for MIC Determination & Disk Diffusion v 9.0
- EUCAST Clinical Breakpoints - Fungi v 9.0
Select the website links below to learn more about BIOMIC V3’s optional testing features:
API, RapID, Crystal. Liofilchem ID Panels
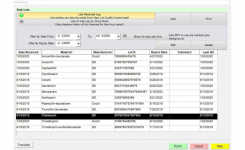
QC & Inventory Management
*96-Well Microtiter - Note in USA: For Research Use Only. Not for use in diagnostic procedures.
Please note : Any products described on this page are
for Research Use Only and not intended for clinical diagnostic procedures unless otherwise stated.