The Importance of Routine Surface Testing for COVID-19
Viruses are spread mainly through close contact between people, although how easily they are transmitted from person to person varies. The new coronavirus disease is highly contagious and spreads more efficiently than other respiratory illnesses such as influenza (the common flu). COVID-19, the disease caused by the new coronavirus SARS-CoV-2, is mainly transmitted between individuals from respiratory droplets produced when coughing, sneezing, or talking.
According to W.H.O. and C.D.C., surfaces and objects are also potential infection routes of COVID-19, as infected droplets can land on them and individuals can be consequently contaminated by touching these fomites and then touching their faces (mouth, nose, or their eyes). Studies have shown that virus particles found in respiratory droplets, can survive on common surfaces for many hours, even days, increasing the risk of infection. Moreover, a study conducted by Australia's national science agency (CSIRO) found that the virus can survive on surfaces such as stainless steel, glass, paper/polymer banknotes and vinyl for up to 28 days, under specific conditions related to temperature, relative humidity and light1.

Positive test result indicates an active contamination risk in the facility, while a negative one confirms the successfully implemented sanitation protocols.
Surveillance and the prevention of cross-contamination are critical in the control of virus spread. Routine surface testing strengthens the existing hygiene measures and sanitation protocols by providing an early indication of contamination by COVID-19 and reassuring the safety of employees, customers, and visitors in various sectors and places. Although surface testing is complementary to existing preventive measures, it is a necessary action that must be taken in order for a company or organization to be fully compliant with COVID-19 safety protocols and to minimize the infection risk.
Equal focus should be given to packages and materials being packaged because even though employees are wearing masks and gloves, there is still risk of contamination.
A major concern is associated specifically with cold-chain foods as the virus can survive for a long time under frozen and cold-storage conditions and consequently spread more easily.
Regarding working places, sanitation measures should be applied on surfaces which come in touch with the naked hand continuously throughout working shifts. Main points of concern are door handles, switchboards, shelves & countertops in the production area, as well as tables, door handles, and shelves.
Over the last months, China has ramped up testing on frozen food after many reported incidences of coronavirus found on imported products and packaging.
The Centre of Disease Control and Prevention (CDC) in the United States say there is a very low risk of infection from handling food packaging, but the Chinese CDC say contact with contaminated packaging could lead to human infection.
Moreover, Chinese officials have linked the recently increased COVID-19 cases in their country to primarily refrigerated imports and people returning from abroad.
Thus, the Chinese authorities have posed very strict regulations by introducing mandatory testing on all international shipments, which consequently causes delivery delays and raises many complaints from the export companies.
In order to speed up the procedure, custom authorities could include the use of rapid surface tests in their testing protocols, that produce real-time results in just a few minutes.

A Novel Approach to Environmental Surface Testing
Rapid Surface Ag 2019-nCov is an easy-to-use rapid test that does not require any special equipment; it can be used on-site and produces accurate results in just 10 mins.
The test is designed to detect concentrations of the virus that pose a risk of infecting individuals who touch a contaminated surface. In addition, Rapid Surface Ag 2019-nCov is a highly sensitive test that can detect virus concentrations 300x times less than what scientists believe is a plausible amount of virus that may be deposited on a surface (according to a reference study this would be 3.38 × 105 TCID50 /ml.*)
Developed by ProGnosis Biotech, it is the first product for surface testing within the COVID-19 diagnostics market to utilize this rapid innovative methodology. Rapid Surface Ag 2019-nCov is a reliable test for companies, authorities and organizations, helping them to monitor and reduce the spread of COVID-19 in their facilities.

Author: Mr. Giorgos Papageorgiou - Co-owner and the Chief Operating Officer (COO) of ProGnosis Biotech
RT-PCR Tests
ThermoFisher Scientific SARS-CoV-2 PCR Workflow for packaging and environmental surfaces is a complete end-to-end workflow, encompassing sampling, sample preparation, and detection. It can deliver results in as little as three hours, quickly providing customers with the information they need to manage any potential surface or packaging contamination risks.
The test, which has been validated for the qualitative detection of the virus on stainless steel, includes three TaqMan RT-PCR assays, to target SARS-CoV-2 (ORF1ab, N-gene, S-gene) genes, and one positive control assay, targeting the Human RNase P RPPH1 gene offering both high specificity and sensitivity. Because of the multi-target design of this assay, overall test sensitivity should not be impacted by the new SARS-CoV-2 strain lineage – B.1.17 variant. It can be used on the Applied Biosystems 7500 Fast Real-Time PCR System or the Applied Biosystems QuantStudio 5 Real-Time PCR System.
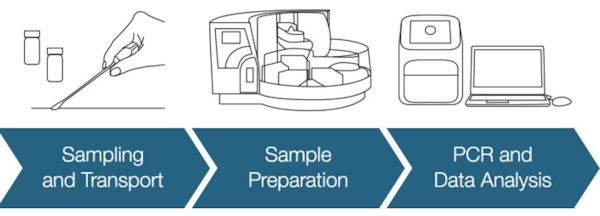
Eurofins GeneScan Technologies' VIRSeek SARS-CoV-2 Solution is a comprehensive workflow for the detection of SARS-CoV-2 on environmental surfaces that includes swabbing of environmental surfaces, RNA extraction, analysis by Reverse Transcription Polymerase Chain Reaction (RT-PCR) and evaluation by an advanced algorithm.
Within the VIRSeek SARS-CoV-2 workflow, the efficient removal of the virus from the swab and automated isolation of high-quality RNA is guaranteed by the VIRSeek RNAExtractor. Following RNA extraction, the lysates are analysed with the VIRSeek SARS-CoV-2 Mplex real-time RT-PCR kit. The kit is highly specific for two regions, N1 and N2, in the SARS-CoV-2 nucleocapsid gene, ensuring reliable results with a two-target approach.
Reference:
1. Riddell, S., Goldie, S., Hill, A. et al. The effect of temperature on persistence of SARS-CoV-2 on common surfaces. Virol J 17, 145 (2020).