Food safety depends in large part on the hygienic conditions in food manufacturing facilities. The presence of high levels of non-pathogenic bacteria can affect the shelf life and quality of foodstuffs for consumers. Equally important is the absence of pathogens (such as Salmonella and Listeria) in food that could potentially cause illness. Food manufacturers must be diligent in keeping their processing environment clean and free of debris to prevent the cross-contamination of the final product.
Yet are current methods, including ATP testing and cultural methods such as agar plates and dip-slides, able to give food producers current, reliable and actionable information about the efficacy of their cleaning and disinfection program?
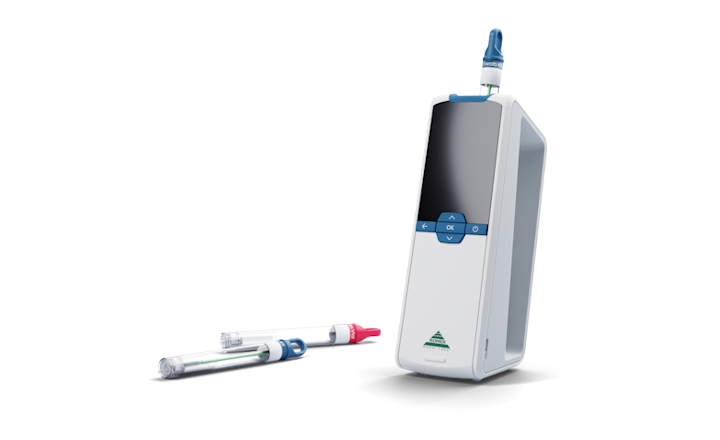
Now, Romer Labs have recently introduced CytoQuant®, the world’s first hand-held, mobile impedance flow cytometer. Rapidmicrobiology talks to Stefan Widmann, product manager for microbiology, Romer Labs to find out why Romer Labs have invested so much time and effort in developing CytoQuant, what makes this instrument a game-changer and why ATP-based testing is not necessarily the best way to verify surface cleaning.
Q: ATP-based testing for surface hygiene is widespread, but what are the downsides?
Stefan: When we test surface hygiene, what we actually want to do is to verify the efficacy of our cleaning and disinfection procedures. Commercial ATP test systems harness the luciferin/ luciferase reaction to generate visible light with the energy provided by ATP. The more light is emitted, the more ATP is present, which could indirectly indicate more food residues. But, while residues are usually a good indication for cleaning efficacy, they are not a meaningful proxy for disinfection efficacy.
Studies like Ching et al.1 (2020) have shown that there is not necessarily a correlation between ATP and living organisms present on surfaces; disinfectants break down the cell walls of animals, plants and microorganisms, meaning that those cells die while their ATP is preserved, resulting in high counts that do not reflect the actual degree of bacterial contamination. With microbiological criteria being an integral part of any HACCP-based procedure, ATP alone is not the ideal parameter of surface hygiene. Used in combination with culture-based methods, the full surface hygiene picture is not available preoperationally, rendering the time-saving advantage of ATP negligible.
Q: CytoQuant - What exactly is it and how does it work?
Stefan: CytoQuant is a mobile flow cytometer that enables the immediate, on-site verification of cleaning procedures in food production facilities or other areas where hygiene is crucial by directly quantifying bacteria and residues on surfaces. In this way, it allows for preventive control and preoperational decisions based on microbiological criteria.
How does it do this? Through impedance flow cytometry, a technology that measures cell and particle characteristics using electrical impedance.
A swab sample is suspended in a conductive solution that is pumped through a microfluidic flow cell with integrated electrodes. An object flowing between the electrodes will introduce a change to the electrical current.
Bacteria have unique electrical properties. Because of their size, the non-conductivity of their cell membrane and the conductivity of their cytoplasm, bacteria leave a fingerprint that distinguishes them from all other particles. This is how CytoQuant can accurately detect and differentiate between intact cells and particles in a sample. The device gives you separate, precise counts for bacteria and residues in 30 seconds.
Q: Flow cytometry? Sounds like it might be a high cost per test. Do you need a skilled operator with full lab?
Stefan: CytoQuant is a mobile, handheld device that is very simple to use. You don’t need a lab or any special training. You sample like you normally would for an ATP test and read the results in 30 seconds. The total cost per test is a fraction of that of traditional fluorescence flow cytometers and very much in the range of what food producers are willing to spend.
Q: How low can it go? What’s the lower limit of detection?
Stefan: Before boiling it down to a figure, we first need to understand what exactly we are measuring and how it relates to currently known parameters. CytoQuant provides precise counts of intact cells and particles. Intact cells are all cells with an intact cell wall, irrespective of their required growth conditions (aerobic or anaerobic, pH, nutrition, salt concentration, temperature, lag-time, incubation time, viable but not culturable, etc.). Because no solid or liquid media can cover this full range, and because only about 1% of existing bacteria are culturable, intact cell counts and plate counts are two very different parameters: results taken from real samples would be expected to deviate from each other.
The devices’ lower limit of detection is 15,000 intact cells per millilitre, a sensitivity which will suffice for most applications.
The device classifies any objects that are not bacteria and that are smaller than 5µm that pass through the microfluidic flow cell as particles. The LLOD for particles is 50,000 per millilitre.
Q: How does CytoQuant distinguish between viable organisms and those recently exposed to sanitiser?
Stefan: The scientific consensus is that the best way to differentiate between viable and dead bacteria is by determining their cell wall integrity. Bacteria exposed to sanitizer ultimately lose their cell wall integrity.
And this is exactly what CytoQuant is developed to detect. It uses the characteristics of an intact cell wall and the cytoplasm within a bacterium to differentiate between intact cells and particles. Unlike cultural-based methods, CytoQuant also detects sub-lethally damaged bacteria.
.
Q: We would really like to see some validation data or user experience. Is there any data you can share?
Stefan: We are currently running several large studies which show how CytoQuant performs in different production environments. Preliminary data are showing promise and are in line with the myriad of lab studies concluded so far. In one of these studies, depicted below, we, working with a university partner, compared the intact cell counts of the CytoQuant mobile flow cytometer with counts from a fluorescence microscope, a fluorescence flow cytometer (Cytoflex, Beckman Coulter) and those from aerobic plates (TSA: tryptic soy agar).
Overnight cultures were prepared by inoculation of tryptic soy broth with -20°C frozen cultures from our strain collection. Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC 25923 was used as reference for a Gram- and a Gram+ bacteria strain respectively . Dilutions in phosphate-buffered saline were made to cover the whole quantification range of both flow cytometers (1E+04-1E+07 cells/mL). Measurements were done in triplicate. Microscope and plates counts were conducted at 1E+06 cells/mL and 1E+02 CFU/mL, respectively. Mean and standard deviations (SD) were calculated.
The results were extremely promising; not only were counts from CytoQuant closest to those of the microscope method, which we can regard as the reference method, CytoQuant also had the lowest average standard deviations of all methods. As expected, tryptic soy agar plates returned the lowest bacteria counts as not all colonies emerge from a single cell and not all cells eventually multiply and form colonies. The fluorescence flow cytometer (Cytoflex, Beckman Coulter) had significantly lower bacteria counts than CytoQuant and the reference microscope method.
Q: Why should QC managers make the switch to CytoQuant now? What’s involved in validation?
Stefan: QC managers need to be able to make informed decisions fast as preventive control and pre-operational actions are not only enforced by regulations and standards but are also crucial to securing food safety. CytoQuant enables them to do just that as it is currently the only solution on the market able to provide a clear picture about the microbiological condition of production surfaces in time. Validating CytoQuant is no different than evaluating any other parameter like CFU or RLU in any production environment. Just like any other risk-based approach, CytoQuant requires a statistically significant number of measurements to define a baseline and the related action limits for specific control points.
Q: Are there applications for CytoQuant in final product testing too?
Stefan: Current efforts are focusing on surface sampling applications. That said, CytoQuant has great potential in areas other than food production. Anywhere hygiene is crucial, such as clean rooms and hospitals, are good candidates to be fields of application.
To learn more about CytoQuant®, visit cytoquant.com or click on the Request Information button below.
[1] Ching, Chai Lay, Arif Kamaruddin, Chandraprasad S. Rajangan, “Assessing the Performance of a Real-Time Total Adenylate (ATP+ADP+AMP) Detection Assay for Surface Hygiene Monitoring in Food Manufacturing Plants and Commercial Kitchens,” Journal of Food Protection, Vol. 84, No. 6, 2021, Pages 973–983.