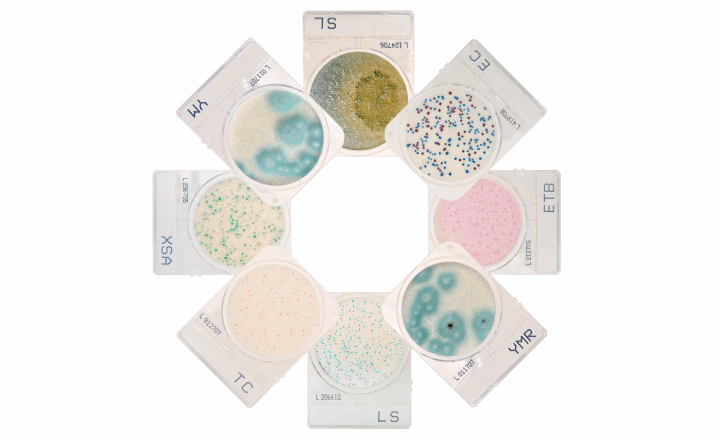
Perfect for screening, confirmation, and more, CompactDry™ is an easy and efficient microbiology solution for labs of every size. Protect your turnaround time and bottom line with our easy to use media system.
The benefits are easy to spot:
- Room temperature storage and longer shelf lives compared to traditional agar media.
- Self-Diffusing media. Most plate types are inoculated directly from primary dilution with a 1ml liquid sample. Through capillary action, the sample diffuses evenly and automatically across the embedded membrane. No need for stamping, spreading, or streaking.
- Sturdy, stackable cassettes ensure that plates will remain stable throughout the process. The friction lid design also protects from the kind of leakage that can be experienced with comparable dry media products.
- Easy to label.
- Designed to work with membrane filters.
- Organism specific media that offers colony counting and chromogenic read, providing both a qualitative and quantitative result.
- Perfect confirmation partner for PCR or other molecular methods.
- SOPs for all CompactDry™ media in the cannabis testing lab available. Simply validate the protocol for easy adoption into your lab.
- AOAC, Microval (ISO compliant), and Nordval validations on most plates. Health Canada approval on Total Count.
- The Wizard™ CompactDry™ automated plate reader boosts lab efficiency and data security when added to the CompactDry™ workflow.
Learn more about Hardy Diagnostics and CompactDry™!