HORIBA UK Limited announces that it is to host an online CPD meeting focusing on ‘Exotic Parasites – The Importance of Testing in The Imported Dog’.
Scheduled for Thursday 1st July 2021 from 19:30 to 21:00 BST, Ian Wright BVMS, MSc, MRCVS, Head of ESCCAP (the European Scientific Council for Companion Animal Parasites) UK & Ireland, will present on the importance of testing protocols in diseases of imported dogs.
Free registration and more information are available through the HORIBA website and by clicking here.
Summary:
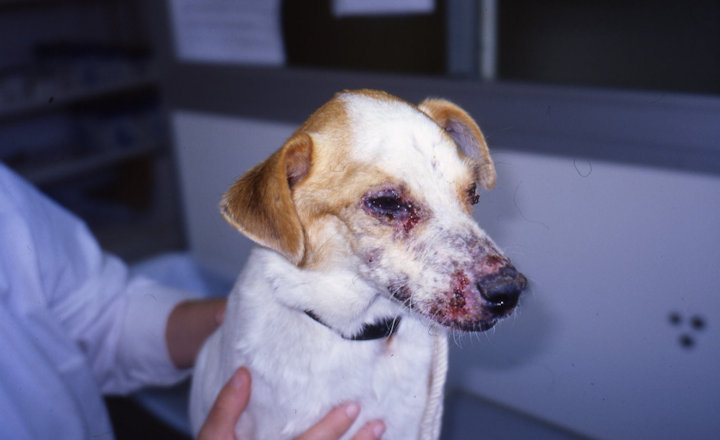
The number of pets imported to the UK has risen sharply throughout the COVID pandemic, with puppy prices reaching levels previously unseen and giving rise to many being imported illegally.
Pets imported via official routes typically undergo a series of detailed tests and prophylactic treatments to prevent the transmission of exotic diseases. However, many of the current tests in place, such as ELISA, are troublesome and laborious, with veterinary clinicians waiting extended periods of time for test results from reference laboratories. This increases the risk of poor outcomes and zoonotic disease transmission.
To help understand and combat this issue, HORIBA is offering an online CPD meeting, delivered by parasitology expert, Ian Wright BVMS BSc MSc MRCVS. The webinar aims to provide education and support to veterinary professionals, such as veterinary surgeons and students, registered veterinary nurses and student veterinary nurses, veterinary care assistants, and reception staff.
Ian Wright is a practicing veterinary surgeon with a master’s degree in veterinary parasitology and is also ESCCAP Guideline Director, an editorial board member for the Companion Animal and VetCPD Journal. Regularly publishing in renowned, peer-reviewed journals in addition to running a veterinary practice, Ian is actively involved in veterinary research, including work on tick-borne diseases and intestinal nematodes.
The webinar-based meeting will provide an overview of emerging veterinary diseases with a particular focus on exotic parasites, discussing the need and emergence of accurate testing protocols and equipment in a time of exponentially increasing imported diseases. A final Q&A session will provide an opportunity to discuss specific questions.
Also to be introduced in the webinar, is a new rapid and easy-to-use, benchtop veterinary PCR system - the POCKIT™ Central* - to help overcome testing challenges and provide an accurate screening tool for the identification of pathogens. Running up to eight samples, HORIBA’s POCKIT Central PCR has a simplified ‘sample in, result out’ process with results available in just 85 minutes, giving veterinary surgeries access to faster diagnoses within their own practice.
For free registration and more information on HORIBA’s online CPD meeting on ‘Exotic Parasites – The Importance of Testing in The Imported Dog’, see the HORIBA website or click here.
*available from HORIBA in selected countries in Europe.