 The antimicrobial combination of ceftolozane and tazobactam is now available on FDA-cleared microbroth dilution susceptibility plates, giving microbiology laboratories the means for susceptibility testing of non-fastidious, Gram-negative organisms that cause complicated urinary tract infections (cUTI), including pyelonephritis, as well as Gram-positive and Gram-negative organisms that cause complicated intra-abdominal infections in adults.
The antimicrobial combination of ceftolozane and tazobactam is now available on FDA-cleared microbroth dilution susceptibility plates, giving microbiology laboratories the means for susceptibility testing of non-fastidious, Gram-negative organisms that cause complicated urinary tract infections (cUTI), including pyelonephritis, as well as Gram-positive and Gram-negative organisms that cause complicated intra-abdominal infections in adults.
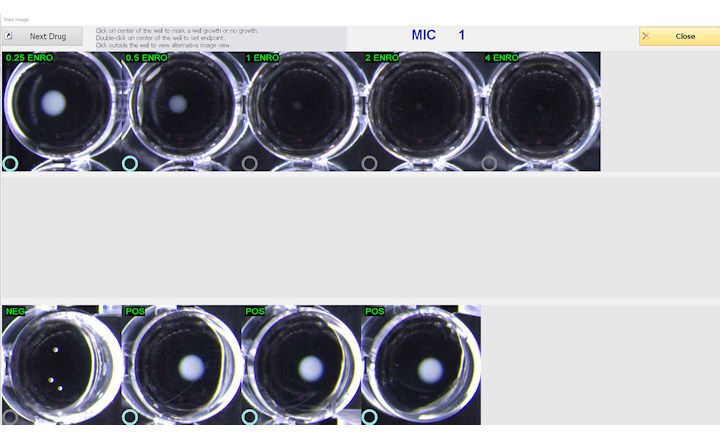
Recognized for having one of the largest, most up-to-date offerings of FDA-cleared antimicrobials for susceptibility testing, the Thermo Scientific Sensititre ID/AST System is now the first to offer ceftolozane/tazobactam on IVD-labeled, microbroth dilution susceptibility plates. The Sensititre System enables laboratories to utilize ceftolozane/tazobactam, along with the choice of over 240 other antimicrobials, to create custom plates tailored to their formulary, dilution ranges and local patient population, eliminating offline tests and reducing costs. Ceftolozane/tazobactam is available globally on Sensititre custom plates, with plans to feature it on new standard plates in the future.
According to Finn Albrechtsen, vice president of R&D and business development, Thermo Fisher Scientific microbiology, “One of the keys to fighting antibiotic resistance is the development of novel antibiotics, and at Thermo Fisher we remain committed to working with pharmaceutical companies to help bring their newest compounds, like ceftolozane/tazobactam, to microbiologists worldwide to provide them with another option in making recommendations for the effective treatment of patients.”
The Sensititre System utilizes true minimum inhibitory concentration (MIC) results, which are essential to understanding antimicrobial resistance, providing greater sensitivity for better resistance tracking. True MIC results are also the preferred measure of antibacterial effect, which can assist with therapeutic choices, and promote overall better patient care.
ZERBAXA® (ceftolozane and tazobactam, Merck & Co., Inc.) is approved by the FDA for the treatment of complicated urinary tract infections (cUTI) in adult patients, including pyelonephritis, caused by the following Gram-negative microorganisms: Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis and Pseudomonas aeruginosa. It is also approved for complicated intra-abdominal infections, in combination with metronidazole, for the following Gram-negative and Gram-positive microorganisms: Enterobacter cloacae, Escherichia coli, Klebsiella oxytoca, Klebsiella pneumoniae, Proteus mirabilis, Pseudomonas aeruginosa, Bacteroides fragilis, Streptococcus anginosus, Streptococcus constellatus, and Streptococcus salivarius.
More information on the Thermo Scientific Sensititre System can be found here, by visiting www.thermofisher.com, or by contacting microbiology@thermofisher.com.