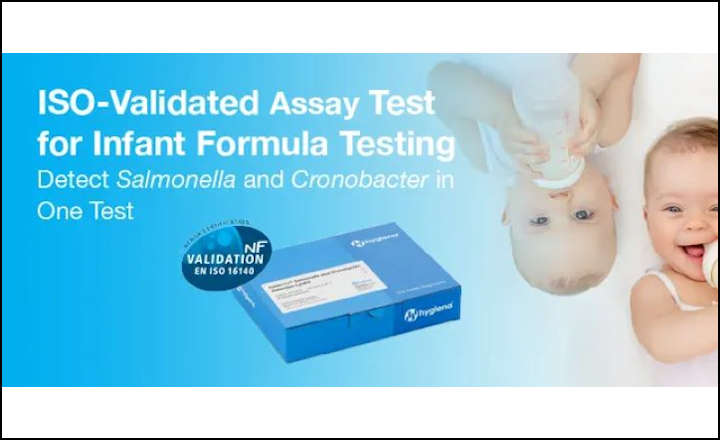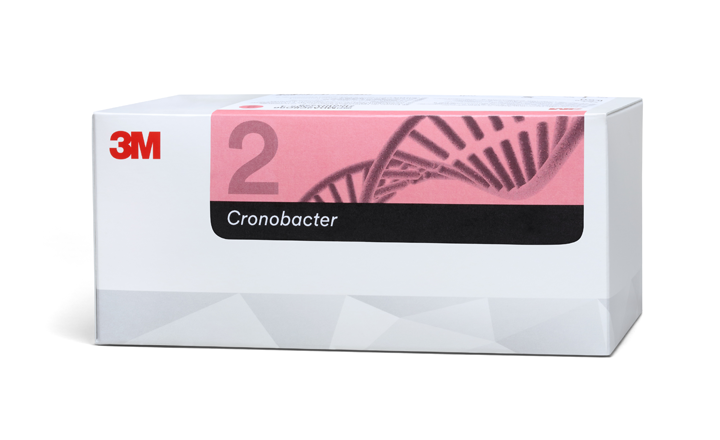
3M Food Safety's 3M™ Molecular Detection Assay 2 – Cronobacter has received two important scientific validations from third party organizations: an international NF VALIDATION certification granted by AFNOR Certification as well as first action status as an Official Method of AnalysisSM by AOAC INTERNATIONAL. The 3M Molecular Detection Assay 2 – Cronobacter provides accurate results in as little as 18 hours of enrichment with fewer steps compared to traditional agar methods.
“Cronobacter is one of the most dangerous pathogens for producers of powdered dairy products, especially for producers of infant formula,” said 3M Global Marketing Manager Carolina Riba. “We’re proud to be able to offer processors a rigorously evaluated test that enables them to obtain results they can feel confident about while saving time and money.”
NF VALIDATION by AFNOR Certification The NF VALIDATION certification process is comprised of two phases, beginning with a method comparison study in which a single, expert lab thoroughly tests the new technology’s effectiveness versus standard methods. Phase two involves an inter-laboratory study where several laboratories compare the efficacy of both the new test method and reference method under defined conditions of reproducibility and repeatability. The NF VALIDATION for the 3M Molecular Detection Assay 2 – Cronobacter (#3M 01/20 - 3/18) certifies that the test is validated for the detection of Cronobacter spp. in powdered infant formula and infant cereals with or without probiotics, raw materials, and production environment samples.
AOAC Official Method of Analysis AOAC INTERNATIONAL facilitates consensus and technical standards for the analytical science communities. Food processor, university, government and contract testing laboratories around the world rely on AOAC Official Method of Analysis validation rigor when choosing chemical, microbial and molecular testing methods. The approval process for becoming an Official Method involves intense review by multiple, independent laboratories and subsequently by an expert review panel. The full study conducted for the AOAC Official Method of Analysis validation of the 3M Molecular Detection Assay 2 Cronobacter (number 2018.01) will be published in an upcoming edition of the Journal of AOAC INTERNATIONAL and will be made available online at eoma.aoac.org
3M Food Safety is a leader of innovative solutions that help the food and beverage industries optimize the quality and safety of their products to enable consumer protection. It provides solutions that mitigate risk, improve operational efficiencies and impact bottom lines.