Liofilchem develops and produces a wide variety of devices for antimicrobial susceptibility and resistance testing.
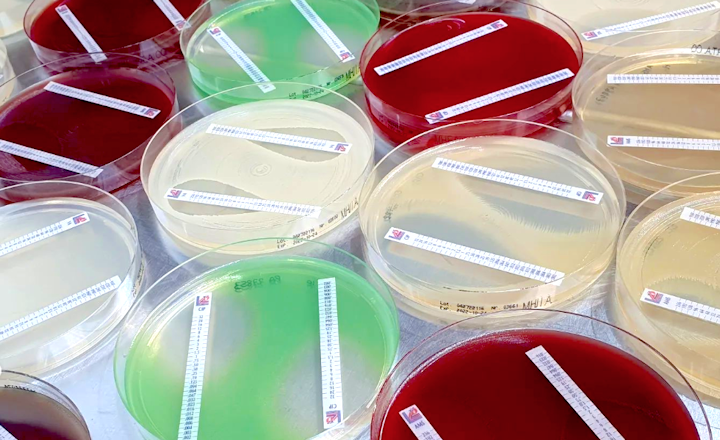
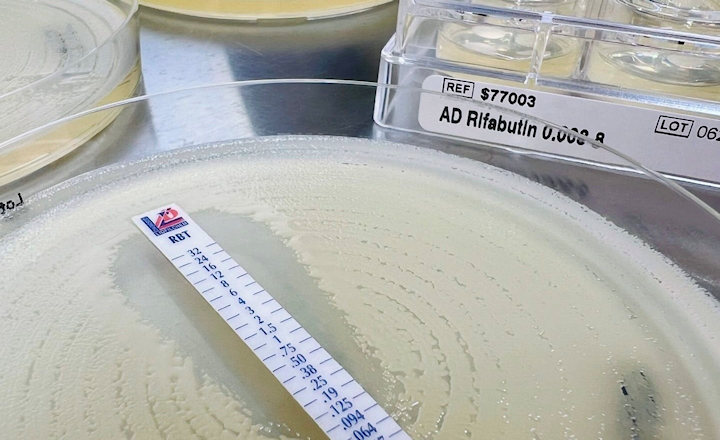
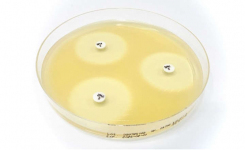
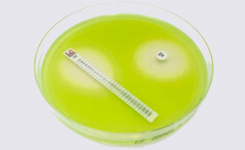
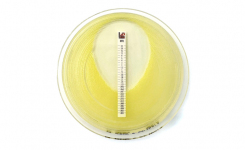
MTS™ (MIC Test Strip) is a quantitative assay for determining the MIC of antimicrobial agents against microorganisms to indicate appropriate patient treatment and for identifying resistance patterns. MTS™ are made of special high-quality paper (European Patent) impregnated with a predefined concentration gradient of antibiotic across 15 two-fold dilutions like those of a conventional MIC method. The MTS™ range comprises over 150 agents. Each agent is available as 10/pack (individually packed), 30/pack (individually packed), or 100/canister pack.
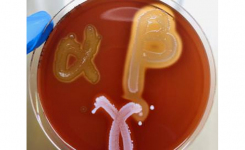
Only MTS™, because of their permeable nature, can be conveniently crossed with the MTS™ SAS (Synergy Application System) (Patent granted in Europe, USA, and Canada) to facilitate and standardize a critical assay such as the in-vitro combination of antibiotics. Antimicrobial resistance is a major challenge for clinicians and clinical microbiologists, and the use of synergy testing is nowadays increasingly requested. MTS™ SAS procedure is significantly faster than any other methods in antibiotic synergy tests (i.e., time-kill, checkerboard), does not require any additional training for those who are skilled in the usage of MTS™, reduces or eliminates the risks of procedure errors when compared to laborious and complicated traditional methods.
Liofilchem produces antibiotic disks and antifungal disks for a total of over 200 agents. Packaging is available as one cartridge of 50 disks, 50 cartridges of 50 disks (250/pack), and a canister pack (250 disks). Kits of antimicrobial agents and resistance inhibitors are available for the determination of resistance mechanisms (ESBL, AmpC, MBL, KPC, OXA-48). Disks are also available as preset or customizable Multodiscs, rings of 8 agents.
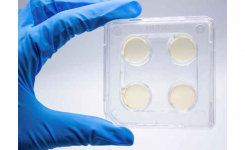
Ready-to-use Agar Dilution panels (Patent pending in Europe, USA, and Canada), available with Mueller Hinton and Fosfomycin or blood agar and Clindamycin, or in customizable versions, are 12-well panels for the MIC determination that can be interpreted according to the CLSI and EUCAST criteria. Panels are individually packed and have four months shelf life.
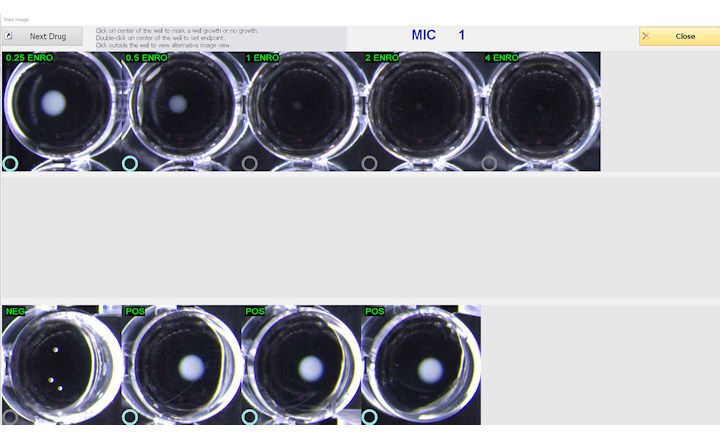
ComASP® (Compact Antimicrobial Susceptibility Panels) are Broth microdilution panels for determining MIC through broth microdilution according to ISO 20776-1:2019 standard.
Produced on a proprietary compact format, ComASP® delivers real MIC results for antimicrobial agents that are indicated by the international guidelines to be used in broth microdilution rather than agar diffusion, agar dilution, or automated/semi-automated methods. ComASP® is also available for customizable assortments of agents and concentration ranges to meet the special needs of laboratories of any size carrying out antimicrobial susceptibility tests. The NP tests are a growing series of panels for the rapid detection of antimicrobial resistance mechanisms.
Rapid ESBL NP® test is an 8-test panel used for the identification of ESBL producers among Enterobacteriaceae. A panel allows the ESBL detection of 8 different isolates. The dried-up substrata in the wells are rehydrated with a lysed microbial suspension. After 20 minutes of incubation at 36±2°C, the result is read and interpreted by a clear color change.
RapidResa Polymyxin Acinetobacter NP® test is an 8-test panel containing desiccated biochemical substrates for rapid detection of colistin-resistant/-susceptible Acinetobacter baumannii isolates. A panel allows the testing of eight (8) different isolates. Test results are evaluated by visual assessment of color development in the wells.
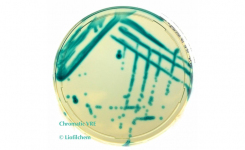
Liofilchem produces a variety of chromogenic culture media for detecting antimicrobial resistance mechanisms: Chromatic Super CAZ/AVI, Chromatic Colistin, Chromatic ESBL, Chromatic CRE, Chromatic OXA-48, Chromatic VRE, Chromatic MRSA, Chromatic Mueller Hinton.
Find out more or contact the supplier for details using the green "Request Information" button below.