Creative Diagnostics has developed a collection of recombinant antigens for SARS-CoV-2 variants, including Spike proteins and Nucleocapsid proteins, covering critical mutations such as K417N/T, E484K, N501Y, and D614G on Spike protein and R203G, G204R, and P13L on Nucleocapsid protein. These reagents can be used to evaluate the efficacy of the antibodies and vaccination.
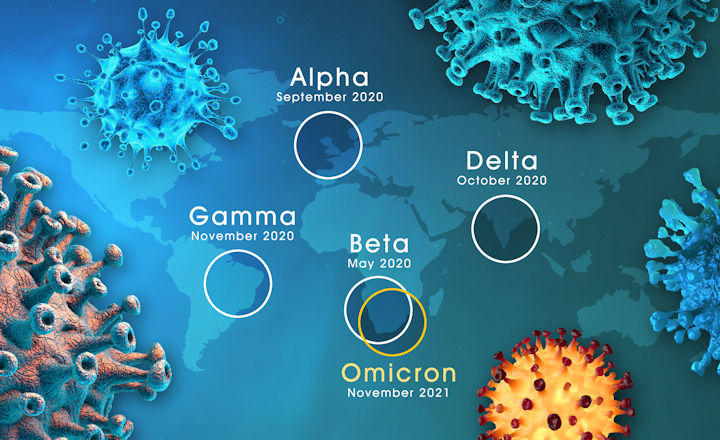
Throughout the COVID-19 pandemic, the genetic variation of SARS-CoV-2 has appeared and spread all over the world. The British variant B.1.1.7, the Brazilian variant P.1, the South African variant B.1.351, the Indian variant B.1.617, and the more recent Omicron B.1.1.529 are particularly worrying because of their high prevalence.
Although there are several mutations in the Spike protein in each variant, three are of particular concern. The N501Y mutation occurs in the receptor-binding domain of the spike protein and is positioned to affect the binding to the host ACE2 receptor. The E484K mutation is found in the South African strain (B.1.1.7) and the strain now is circulating in Brazil (P.1). There is increasing evidence that this mutation may weaken the neutralizing effect of certain antibodies. K417 was mutated to N in the B.1.351 lineage, and it was changed to T (K417T) in the P1 lineage identified in Brazil.
Mutations in these strains also occur in the nucleocapsid protein, which is often used as a biomarker in rapid antigen testing. It is important to evaluate whether current commercial antigen tests can detect mutant N protein with the same sensitivity and specificity as the WT counterpart.
Creative Diagnostics is working hard to develop a series of recombinant antigens for these variants, which can be used to evaluate the efficacy of antibodies and vaccination. A series of recombinant mutants from the Omicron variant B.1.1.529 are also available at Creative Diagnostics for researchers to study the Omicron variant.
For more information about the SARS-CoV-2 Mutants Protein or any other SARS-CoV-2 research products, please visit Creative Diagnostics at www.creative-diagnostics.com or ask for a quote/send an inquiry via 'Request Information' button below.