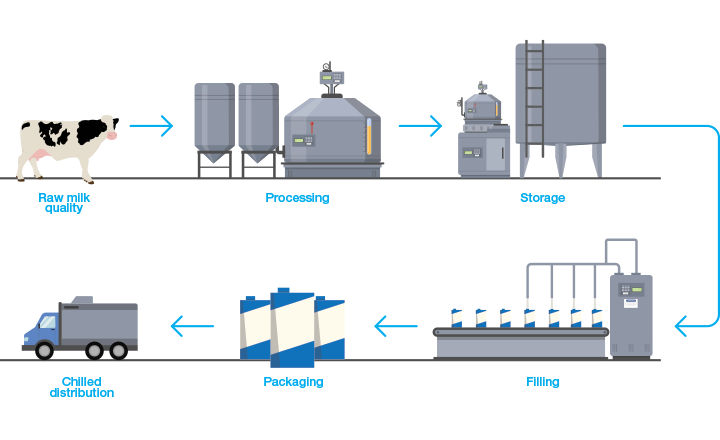
The global aseptic food and beverage packaging market is expected to reach $35 billion by 2025. partially due to the increased popularity of dairy products. This has fueled the demand for products with longer shelf lives and room temperature storage. While ideal for many foods and beverages, aseptic processing poses both advantages and disadvantages for milk and milk-derived beverages.
Milk products are generally required to be processed by thermal methods such as pasteurization, as has been done for decades, which limits shelf life, chilled distribution, and refrigerated storage after purchase. Plus, the shelf life and quality of pasteurized milk can be affected by spore load in the raw milk, pasteurization conditions, contamination, and particularly distribution temperature.
To overcome these challenges, new processing methods are being evaluated to further reduce or eliminate microbes that can cause food spoilage and disease. Two thermal processing methods are UHT (Ultra High Temperature) and UP (Ultra-Pasteurization). While UHT milk has been around for a while, it is now making a resurgence along with Extended Shelf Life (ESL/UP) milk, the latter playing an important role in the dynamics of dairy markets as they develop new processing and packaging methods.
What is UHT processing?
This method involves processing milk at high temperatures (135 – 154°C) for short holding times (1-2 seconds) to achieve what is termed “commercial sterility”. The final product incubates at 55°C for 7 days and at 30°C for 15 days, followed by bacterial growth testing. When combined with aseptic packaging and hermetic sealing, the final product can be shelf-stable (ambient temperatures) with a shelf-life of 6 to 12 months.
What is ESL processing?
ESL processing lies somewhere between pasteurization and sterilization. There are no specific temperature-time requirements for this type of processing in most countries. However, in the US, ESL milk, designated as “ultra-pasteurized”, must be produced by heat treatment of >138°C for >2 seconds (some products are processed from 120°C-140°C for 0.5-4 seconds). Products have reduced microbial counts compared to pasteurization but are packaged under hygienic conditions (not sterile). They have an extended shelf life (45-120 days) under refrigerated conditions, depending on the product and the packaging process.
The Advantages of UHT Milk Include:
- Less processing time
- Extended shelf life
- Room temperature storage
- Reduced microbial load
- Safer than other milk
- Cheaper/wider variety of packaging options
Disadvantages Include:
- Milk quality/altered flavor
- Decreased nutritional value
- Expensive process - highly technical equipment
- Anonymous milk origin (combining multiple supplier batches)
The Advantages of ESL Milk Include:
- Similar to pasteurized milk (taste, smell, color)
- Extended shelf life
- Less expensive than UHT processing
Disadvantages Include:
- Potential for contamination
- Potential for flavor issues
- Texture and appearance changes after prolonged storage
- Difficult to remain competitive
The best solution?
Both processes can be optimized, depending on the final product. No matter what process you choose, microbiological testing is required. While each type of milk has specific testing requirements, both will be considered adulterated if they contain Salmonella species, EHEC O157:H7 and other enterohemorrhagic Escherichia coli, Campylobacter jejuni, Yersinia enterocolitica, vegetative cells of Clostridium botulinum, Clostridium botulinum toxin, Staphylococcus enterotoxin, or Bacillus cereus enterotoxin. Also, a product cannot contain levels of non-toxigenic E. coli above 10 MPN (most probable number) per gram in two or more subsamples or greater than 100 MPN per gram in one or more subsamples. For other organisms, levels must be below 10,000 CFU/gram.
A single, rapid solution

The Innovate platform features RapiScreen™, ATP bioluminescence technology, the industry standard for screening dairy products due to its speed and clear absence/ presence results. ATP testing eliminates the need for plate incubation, allowing products to be released days faster. The system can run 96 individual samples at once with no secondary incubation required. This is possible due to a unique technology that eliminates somatic cell (non-microbial) ATP and allows testing products with a wide range of pH values.
The Innovate System is ideal for UHT and ESL products as they should be either free of contaminants or contain low levels (less than 10 CFU/ml). In addition, the RapiScreen™ reagents have been validated on raw materials, in-process formulations, and finished goods. The automation allows simultaneous testing of 96 different samples on a single microtiter plate every 30 minutes. Subsequent assays can be prepared even while the system is in use to keep high-volume operations running smoothly.
Other testing methods either rely on pH or plate culture. Measuring pH requires longer incubation times and often misses some microorganisms as some dairy formulations are buffered, minimizing pH shifts. Standard plating methods require different enrichment media, long incubation times (7-14 days), and long detection times (2-5 days) to target contamination.
The Innovate System was developed to simplify workflows by detecting all viable microorganisms (aerobic, anaerobic, and yeast/ mold) in a single test. Although sample product pre-incubation may be the same as plating and pH methods, Innovate can achieve same-day “detection”. The Innovate System can be adapted to determine “pass” or “fail” criteria, making it even easier to monitor testing - green is pass and red is fail. The system also enables sample data from multiple instruments to be saved to a single database on the company’s secure network, helping simplify regulatory compliance. Data can be securely viewed onsite or remotely, and reports can be generated for analysis.
When utilizing the rapid screening kit for dairy on the Innovate System, dairy processors can minimize costs for warehouse space or holding product while feeling reassured that any contamination events will be identified early. In addition, the Innovate Autosampler III can be paired with the Innovate platform to prepare up to 2,000 assays per hour for analysis in the Innovate System, simplifying the workflow and freeing up valuable technician time.