Specific Diagnostics has announced CE-IVD registration of the Reveal rapid antimicrobial susceptibility testing (AST) system. European availability of Reveal is now beginning in France, led by 6 distinguished evaluation sites.
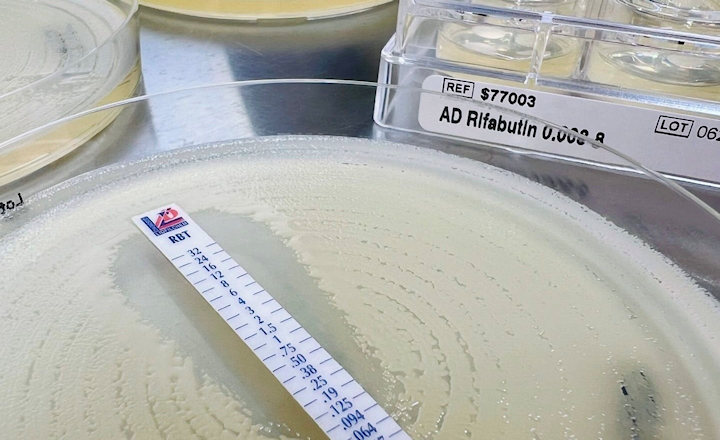
The system provides minimum inhibitory concentration (MIC) with wide antimicrobial coverage for bacteria species responsible for bloodstream and other acute infections.
Current methodologies require 2 days to determine the effective antibiotic, while Specific's Reveal AST solution provides antibiotic susceptibility results in hours, allowing same-shift impact.
“Reveal’s same-shift AST, with an average time to result of 4.6 hours based upon our recent clinical study, is much-needed for time-sensitive infections, while the system’s affordable cost, ease of use and high throughput is compatible with the requirements of hardpressed microbiology laboratories”, said Dr Paul A. Rhodes, CEO of Specific.
“We have been pleased to work closely with Specific as they have developed manufacturing under the formal quality system appropriate to medical devices in Europe,” said Mr Maurizio Suppo, co-founder and Partner at Qarad, Specific’s Brussels-based Registered Agent in Europe.
Specific intends to offer Reveal for evaluation and clinical use in France and the United Kingdom during Q4 2020 and Q1 2021, with availability in additional markets thereafter.
Note: This content has been edited by a rapidmicrobiology staff writer for style and content.