The qPCR reagents designed by the CDC to test for SARS-CoV-2 has scored 100% in a study carried out by Dr Gary Procop, board member of the American Society for Clinical Pathology (ASCP). Dr Gary Procop, who leads COVID-19 testing at Clevland Clinic, Ohio, also recorded over 96% accuracy for the Roche cobas SARS-CoV-2 test and 98% accuracy for the Cepheid point-of-care Xpert Xpress SAR-CoV-2 test.
The study, which has yet to be peer-reviewed and published, was performed by Procop and his team at the Cleveland Clinic, where they took 239 patient samples known to be positive for the virus and re-tested them using five products available through the US Food and Drug Administration’s (FDA) emergency use authorization.
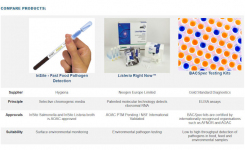
The other FDA-EUA tests studied were the ID NOW™ point-of-care test from Abbott Diagnostics, which gives positive results in 5 mins and the Simplexa COVID-19 Direct test from DiaSorin Molecular, that has a one-hour sample-to result turnaround. The study found an unacceptable false-negative rate in both of the tests, with the DiaSorin test having a false-negative rate of 11% and the Abbott test at nearly 15%.
Abbott responded to the study, claiming a dilution must have occurred when viral transport medium (VTM) was used. Abbott has now changed the Instructions for Use (IFU) under its FDA-EUA to instruct users to place swabs directly into the device without VTM.