Bruker have acquired a technology platform and product line that will further expand its range of bacterial, viral and toxin detection solutions for homeland security bio-detection and for selected microbiology applications. Financial details were not disclosed.
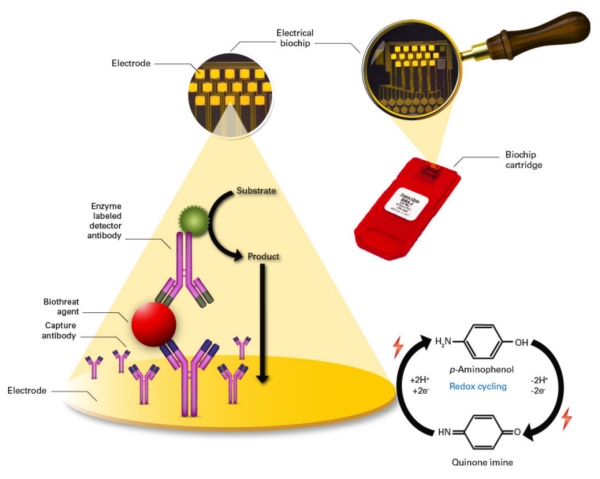
The acquired assets include intellectual property, a field-portable, electrochemical chip-based detection instrument and associated consumable kits for on-site, fast identification of selected bacteria, viruses and toxins.
Detection and identification of up to 6 bio-threats per assay chip occurs typically within 20 minutes, with excellent sensitivities and without any prior culturing steps for bacteria. The immunoassay kits, which Bruker now can offer for bio-detection and homeland security, include:
Biothreat Test: Bacillus anthracis, Yersinia pestis, Francisella tularensis, Brucella species, Burkholderia mallei/pseudomallei, Orthopox viruses (smallpox);
Toxin Test I: Botulinum Toxins A, B, F; Staphylococcal Enterotoxin B; Ricin;
Toxin Test II: Botulinum Toxin C, D, E; Staphylococcal Enterotoxin A; Abrin;
The acquired instrument platform, the portable Bio-Detector integrated (pBDi), is ready for immediate commercial launch. Unlike other PCR-based field methods, the unique multiplex ELISA-based detection with a novel electrochemical chip readout, allows rapid on-site identification of various bio-threats, including toxins, with very high sensitivity.
Developed for ease of use by non-scientific personnel, the pBDi can even be operated while working in protective gear under field conditions. Fully portable and operating from internal batteries, the pBDi can be used in 'hot zones'.