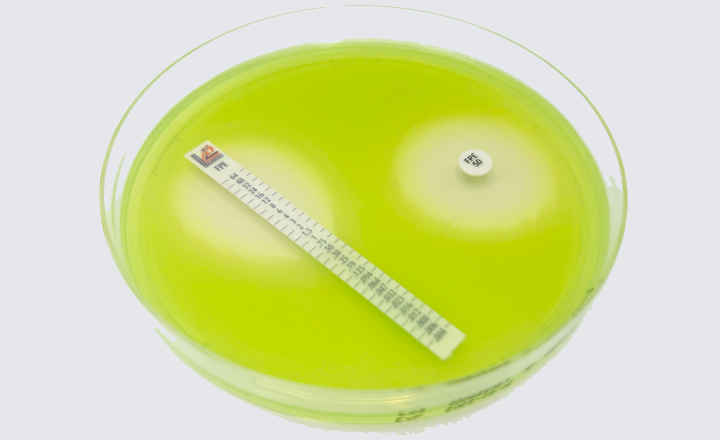
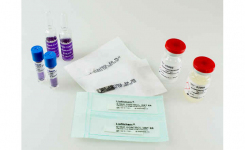
Key Points:
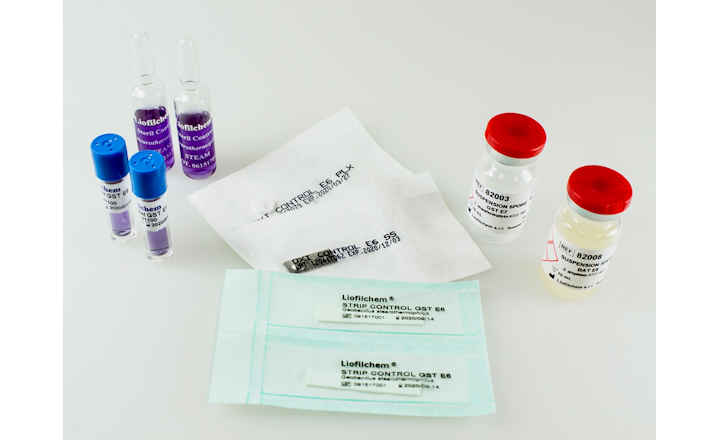
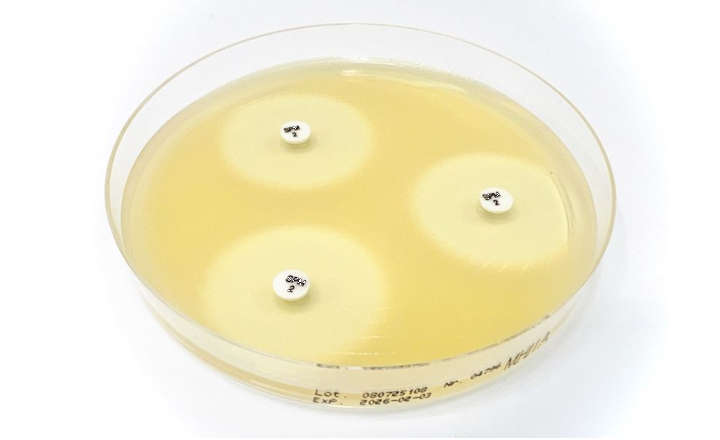
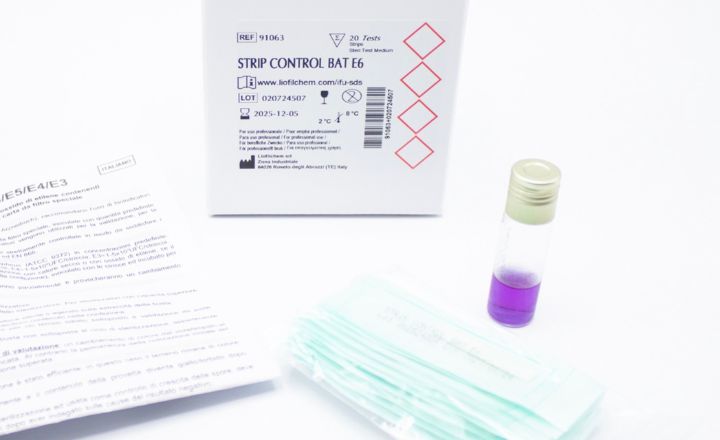
- Bioindicators manufactured by Liofilchem are available in ampoule, self-contained, strip, suspension and coupon formats in several materials such as glass, aluminum, poliflex, steel, PVC.
- We also produce custom-sized Biological Indicators on request.
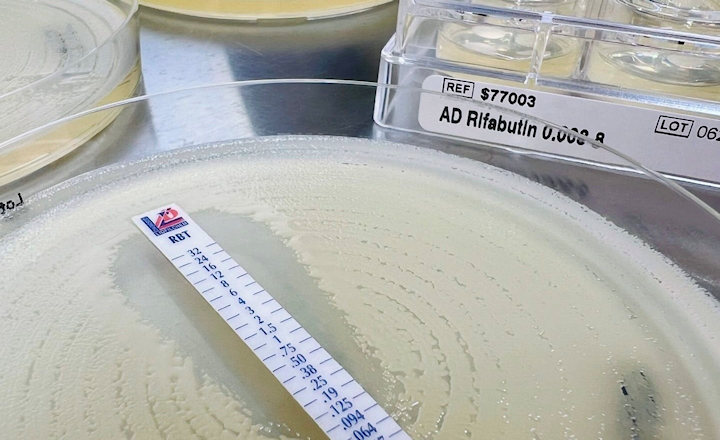
- All our Bioindicators have a certificate reporting D values, survival time, kill time, population and purity.
- Biological Indicators are manufactured in strictly controlled environment.