With microbiological quality control in a food or pharmaceutical processing plant, every hour spent without taking action can be critical. In a global supply chain, product recalls can involve multiple countries, create financial losses and damage brands’ reputation.
Internal and outsourced contract testing microbiology labs both play a key role in protecting businesses and public health from issues due to microbiological spoilage and pathogens. For them, the urgent requirement is to automate, in order to provide more reliable, traceable and accurate test results with a faster turnaround from sample to result.
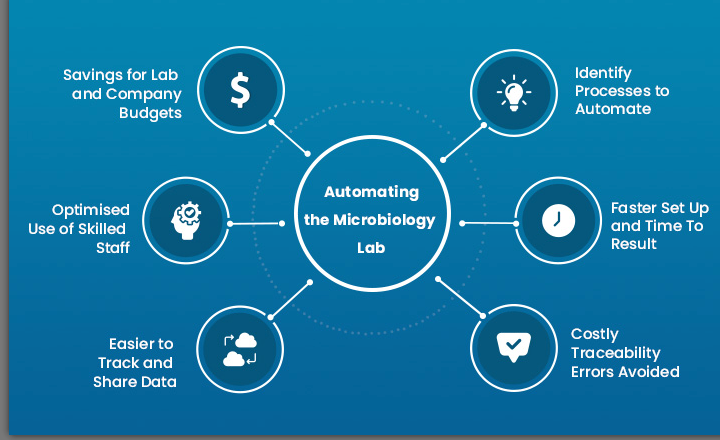
General microbiological analyses, such as total viable counts are ideally suited for automation. From sample preparation to colony counting, the process is made up of several repetitive tasks that require consistent precision. Doing them manually is – first of all - time consuming and secondly prone to human error. The benefits of adopting automation, however, are not limited to faster and more accurate results.
Automated systems can:
- Improve consistency and repeatability. Even with precise procedures in place for each task, it is almost impossible to achieve consistency manually. Small differences will build up at each step of the analysis, resulting in significant variations between similar tests. Mechanical arms and electronic dispensers will use millimetric precision regardless of the volume of work.
- Avoid costly traceability errors. Data collection is another example where a single human error can create serious problems. Modern prep stations assign barcodes to each sample and collect data without errors or losses.
- Save money on consumables. Automatic dispensers lower costs for consumables by avoiding spillages or excessive use of agar.
- Optimize the use of skills. Automation frees skilled lab technicians from tedious and mentally draining tasks, allowing them to use their skills to make better judgment calls. For companies and labs, that also means a more efficient use of valuable human resources.
The Business Case for Lab Automation
The benefits of automation apply to both internal and outsourcing labs, with one important difference: for the first, microbiological analysis supports the company’s core business, while for the latter the core business is the microbiological analysis itself.
This difference creates different mindsets: “For companies, the drive to adopt automation is part of risk management and brand protection, and testing is seen more as an operational expense,” says Luke Cohen, marketing manager at Copan Diagnostics. “For contract labs, automating is a way to provide a better service, cut expenses, and to differentiate themselves from other contract labs - there's a lot of competition in product testing.”
On the other hand, although companies see automation mainly as an operating cost, their investment tends to be lower: “In general, outsourcing labs need to be better equipped to do analysis on different bacteria and provide tests results to different customers. For internal labs, analysis is limited to the type of production of the company, so they don’t need to be as well equipped. Both of them expect automation to integrate in their existing workflow.” says Suzanne Jalenques, head of communication at Interscience.
Automation Examples
Two examples of state-of-the-art technology are the CYCLONE™ from COPAN and INTERSCIENCE’s ScanStation.
The CYCLONE™ is a sample processor for food, cosmetics and beverages, or any substance that comes in contact with the human body and has to be tested for microbiological contamination. It fully automates the traditional pour plating, spiral plating, streaking and spreading petri dish plate preparation methods. It can handle multiple media seamlessly and comes with built-in dilution and labelling capabilities. The benefits for labs are a much more efficient use of agar (or other media) and lower labor and consumable costs. The CYCLONE™ can be integrated with a smart incubator which automates incubation and imaging and uses artificial intelligence (AI) for colony counting and morphology recognition.
ScanStation is an automated incubator and colony counter that monitors up to 300 petri dishes in real time and starts counting colonies from the beginning of incubation. Results can be obtained after only 6 hours, much earlier than the end of the incubation period, which typically lasts 22-24 hours. Receiving reliable, actionable insights much earlier means that agrifood, pharmaceutical or cosmetic companies can release production batches much more quickly if results are negative, or reduce production loss if contamination is confirmed.
The Future of Automation
For companies, automation is a way to protect themselves from the damages caused by contamination incidents: product loss, recalls, and a damaged brand reputation. For outsourcing labs, it is a strategic choice to increase revenue. Technology has not reached its full potential yet, so we asked our experts what – in their opinion – the labs of the future look like.
“The future is not a lab without people,” says Jalenques. “Automation will bring more speed and accuracy and better traceability. There will be much less need for manual intervention, but labs will still need skilled technicians. Their microbiology expertise will still be crucial.”
“Traditionally, there’s always been a clear separation between automating repetitive tasks on one side and human decision making on the other,” says Cohen. “In the future we’ll see AI helping more and more lab technicians in evaluations. AI, however, won’t be actually replacing those decisions. Ultimately, they’ll still be made in the same way. Just with more advanced tools which allows us to process analyses in faster, more efficient ways.”