The biotech giant Thermo Fisher has gone micro at this year’s AACC in Anaheim, California. Among the stacks of new medical devices they have added to their portfolio which include a new HLPC system and two new mass spectrophotometers rapidmicrobiology have picked out the ones relevant for you.
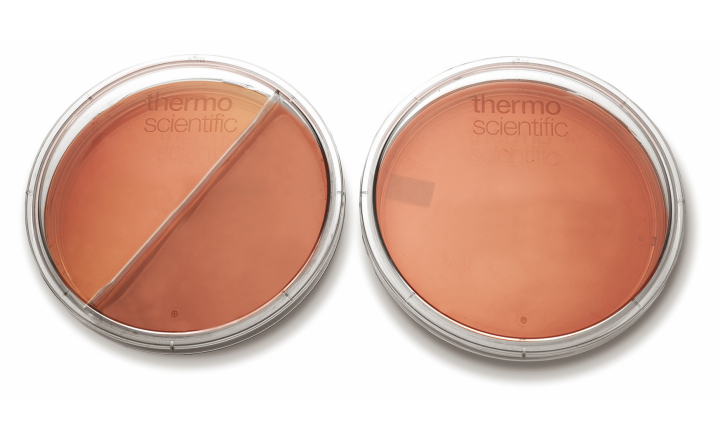
SmartPlate: Re-Inventing the Petri Dish
The dawn of a new era as the petri dish is being reinvented to be compatible with the automation period we are now heading into. The new plate, developed by Thermo Fisher Scientific is an easy to handle hybrid of days gone by and days to come as it can be used for manual and automated methods for the analysis of microbiology samples for clinical, food safety testing and pharmaceutical laboratories.
Improvements made with the SmartPlate allow for easy transport in the laboratory. The SmartPlate comes equipped with stacking rings to enhance stackability and ease of transport for large number of plates in a single movement, so no more balancing acts. By using this new stacking capability laboratories can maximize space in refrigerators and work spaces avoiding clutter and the inevitable mistakes.
The SmartPlate is fully compatible with all available automated specimen processors and is a re-invention of Thermo Scientific’s prepared culture media, notably their Oxoid and Remel prepared media. However this time it reduces the amount of resin normally used in the manufacturing of the plates and so minimizes laboratory waste.
ImmunoCAP Allergen Components Test for Furry Animals Allergen
A new addition to the suite of ImmunoCap assays, which check the immunoglobulin E (IGE) in your blood for sensitization to a particular allergen, is the ImmunoCAP Allergen Components test for Furry Animals. The test which has just been cleared by the US FDA for in vitro diagnostic use will be showcased by Thermo Fisher at the AACC this week. This molecular allergology test will check a person’s sensitivity to specific proteins found in the skin, fur and saliva of dogs, cats and horses. The ImmunoCap assay is able to test if a patient is sensitized to 11 different allergen component proteins found in these animals. From an allergen source, single allergen components can be produced and then it is possible to check if a patient has sensitization to these components individually, which in turn will help to pinpoint on an exact molecular level which component the patient is sensitized to.
Professor Andrew Liu, MD at the Children’s hospital Colorado remarked on the product today in the media, "Since cats, dogs, and horses produce many different molecules to which we can become allergic, component testing can help distinguish if they are contributing to allergy and asthma symptoms better than whole allergen testing via skin prick or blood testing alone. For patients, determining which of the major allergen components they are allergic to can help their healthcare provider be more precise in their diagnosis and management recommendations. When coupled with whole allergen testing, ImmunoCap Furry Animal Allergen Components deliver the information clinicians need to better diagnose and treat patients with pet allergies and asthma”
Thermo Fisher’s Microdilution ARIS HIQ AST
An industry-wide stewardship on the reduction of antibiotic use has seen a wave of new antimicrobial susceptibly tests (AST) being brought to the market. Thermo Fisher’s new Sensititre™ Aris HiQ™ AST system which received approval for use in Europe last month and soon to be launched in the US will be on show at the AACC. This instrument, which relies on microdilution, provides clinicians with accurate minimum inhibitory concentration (MIC) allowing critically-ill patients to be treated with an effective antibiotic.
This benchtop automated reading and incubation system has an expanded capacity of up to 100 Sensititre plates in a limited footprint that makes it possible to process more tests at one time while conserving valuable bench space. It also features an intuitive touchscreen user interface for convenient operation; 24/7 access to critical test information; and batch load and unload capability for improved testing workflow.
Methods like Sensititre’s microdilution and the Epsilometer test are commonly used in laboratories in tandem however neither are recommended by the Clinical and Laboratory and Standards Institute (CLSI) and it is agar dilution listed as the gold standard. In a study published recently in ‘Pathology’ both Sensititre and the Etest were compared and both were given high praise however in this study it was the Etest that was recommended for routine diagnostic laboratory usage.
Compare Your QC Anomalies MAS™ LabLink XL
MAS™ Controls has been Thermo Fisher Scientific’s stable quality control set for a number of years and at this year’s AACC they will showcase new additions to these clinical chemistry and immunoassay standards. Along with their cloud service LabLink XL quality assurance software, clinical scientists and QC can now measure their results with their peers on the LabLink cloud and receive instant access to their peer’s data. If any anomalies show up in data, the problem can be narrowed down by seeing other QC staff’s results worldwide within the cloud. The service also provides troubleshooting capabilities.
Note: This content has been edited by a rapidmicrobiology staff writer for style and content.