Manufacturers of cosmetics and personal care products have a responsibility to ensure that the products they market are safe and not contaminated with harmful microorganisms. Whilst cosmetics do not need to be sterile they do need to be made under hygienic conditions. So what resources are available to manufacturers of cosmetics and personal care products to help them ensure compliance with existing standards? In this special focus we are delighted to include contributions from Liofilchem, Charles River, Sy-Lab, Promicol and Microbiologics.
 |
Charles River Microbial Detection & Identification for Consumer Care Manufacturers Since cosmetics and personal care products are FDA-regulated, manufacturers are held accountable for the safety and quality of their products. Charles River offers microbial solutions to help the consumer care industry comply with best practices for QC. more...
|
|
Liofilchem's Solutions for Cosmetic / Personal Care Microbiology Liofilchem produces TAT Broth, EUGON Broth LT100, Tryptic Soy Broth + Capitol 4 and other culture media customizable in formulation, volume and packaging formats, for cosmetic / personal care microbiology. more.. |
|
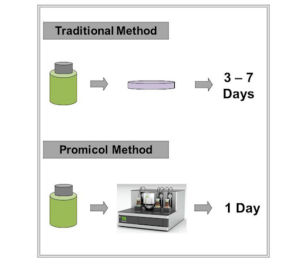 |
ATP Bioluminescence Rapid Testing of Cosmetic Products Using simple test procedures with no change in sample preparation, Promicol’s cosmetics software provides unique security and simple automated control of the complete assay - capable of running approximately 100 samples in 10 mins.
|
|
Rapid Microbiological Results in Full Compliance with ISO 17516 Combined testing using the BacTrac 4300 technology for TMAC and Y+M and the RiboFlow® biomolecular lateral flow assay for E. coli, P. aeruginosa, S. aureus and C. albicans can facilitate and speed up cosmetic testing significantly. |
|
 |
Microbiologics EZ-Accu Shot™ quantitative microorganism pellets, designed specifically for growth promotion testing, provide 10-100 CFU per 0.1 ml of hydrated suspension as required by the USP. Visit the Microbiologics team at booth 207 to learn more. more...
|
|
Real-Time Incubator and Colony Counter Delivers Results from 8 Hrs The award winning ScanStation automatically counts colonies on plates as they are formed, with results possible in 8 hrs. Intended for food, pharma and personal care labs, the system delivers faster, objective results whilst using standard methods.more... |
|
Microbial Testing and Quality Control in Cosmetic Manufacturing Webinar With an increase in regulatory oversight on the horizon Cosmetics Manufacturers will benefit from attending this informative Webinar about Microbial Testing and Quality Control in Cosmetic Manufacturing.more... |