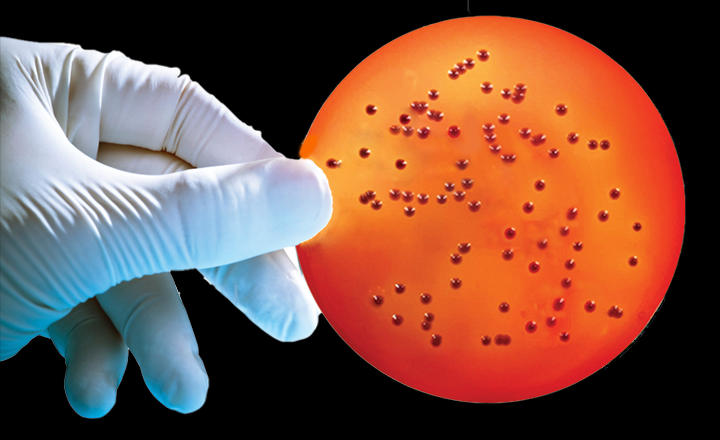
SGL’s Burkholderia Cepacia Selective Agar (BCSA) in 90mm plate format is a selective growth medium designed to meet the formulation requirements listed in USP <60> to recover B. cepacia and BCC.
Impact of Burkholderia Cepacia on Pharmaceuticals
Bcc is one of the contaminants most frequently encountered in sterile and non-sterile pharmaceutical products.
Healthcare-associated outbreaks of BCC have been linked to contaminated nebulized and intravenous solutions, including commercially distributed products, skin care products and disinfectants.
In the past decade, organisms that fall within the BCC grouping have been identified as potentially ‘objectionable’ within the pharmaceutical manufacturing environment.
Testing for Burkholderia Cepacia
With the creation of the USP <60>, the pharmaceutical industry had taken a definite step towards addressing the public health risks posed by B. cepacia.
- Do you test for the presence of Burkholderia cepacia or Burkholderia cepacia complex (BCC) in the pharmaceutical or cosmetic industry?
- Struggling to make a suitable growth medium to detect B. cepacia?
- Unsure which test method to use?
- If you need to test for Burkholderia cepacia in line with USP <60>, let SGL help you with a pre-poured plate media solution.
SGL BCSA pre-poured media contains numerous nutritional components, energy sources supporting BCC growth, and a mixture of antibiotics designed to prevent the growth of non-fermenters not belonging to the BCC and typical environmental isolates such as Pseudomonas and Staphylococcus species.
This allows for easier detection of BCC in test samples containing background contaminants.
Why use SGL pre-poured BCSA?
- Meets the USP formulation criteria.
- Quicker and better isolation of BCC
- Increased Selectivity
- Fewer false positive
- Saving laboratory time
- Consistent media every time
SGL supplies a wide range of other pre-poured media for use in pharmaceutical, cosmetic, clinical and veterinary applications. These include standard off-the-shelf media as well as bespoke media products tailored to customer-specific formulations.
Representative samples of our products are QC tested in our on-site laboratory which is UKAS accredited to ISO 17025.
For more information, contact us directly by using the Request Information button below or visit our website.