Endotoxin testing is not often thought of as a “rapid method”, however, with the advent of pre-spiked plates and a result in 20 minutes, the “EZ II GO Plate” is definitely a quick and easy test, that can significantly improve efficiency. The most time consuming and difficult part of most Endotoxin tests is the preparation of reference standard Endotoxin (RSE). The days of vortexing and diluting standards, and straining your eyes by pipetting tiny positive product control (PPC) spikes into empty wells are over. And with this type of convenience comes the removal of several previous concerns including the following:
1. Did I prepare the curve correctly?
2. Was the vortex time sufficient?
3. Did I plate the standards in the correct order?
4. Did I add the spikes to every appropriate well (it’s hard to see)?
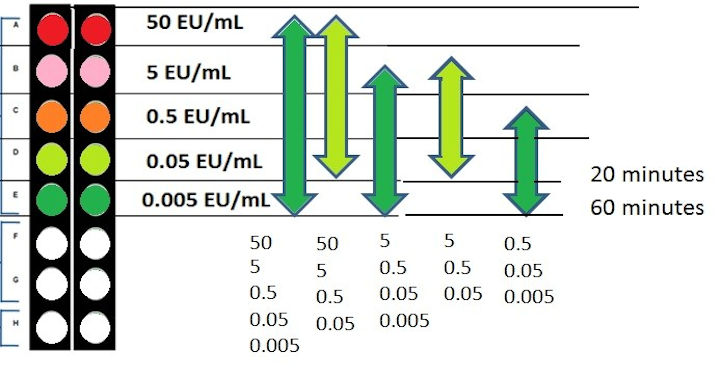
For a 20 minute test, the sensitivity is 0.05 EU/mL which is more than sensitive enough for purified water testing (the USP limit is <0.25 EU/mL).
How easy is it? Take a look at the graphic below. There are three steps, then just hit “Go”.

The best part, in my view, is that with the pre-spiked plate one doesn’t have to worry about having either a standard curve “out” or a poor %CV for standards or products. The precision added and dried reagents make testing a “sure thing”. It is often said "that the most expensive test you will ever do is the test you have to do twice" and this is a problem that is alleviated by using the Go plate. The pre-spiked curve and positive product controls have all been validated and each batch goes through a thorough quality control process to make sure your results are correct.
If you need more sensitivity, you can always take the pre-dried curve out to 0.005 EU/mL as shown in the graphic below. It’s all already dried into the plate, reconstitute all the wells with reagent water and use the software to pick your curve of choice. Thus, not only is it rapid, it’s very flexible.
 About the Author
About the Author
Kevin Williams graduated from Texas A&M in 1982 with a BS in Microbiology. From there spent 30 years at Eli Lilly in Indianapolis. After retirement, he has been at Hospira, Lonza, GE, and now bioMérieux.