Representative of the company’s commitment to offering a food industry-leading test portfolio, Hygiena’s InSite®Salmonella test device has recently received AOAC INTERNATIONAL Performance Tested MethodsSM certification. Hygiena has repeatedly demonstrated its ability to deliver innovative diagnostics and software that meet global food safety requirements while making the testing easier to do, providing faster results and at an affordable price. This latest AOAC RI approval for InSite Salmonella demonstrates the method’s sensitivity, reliability and matrix robustness.
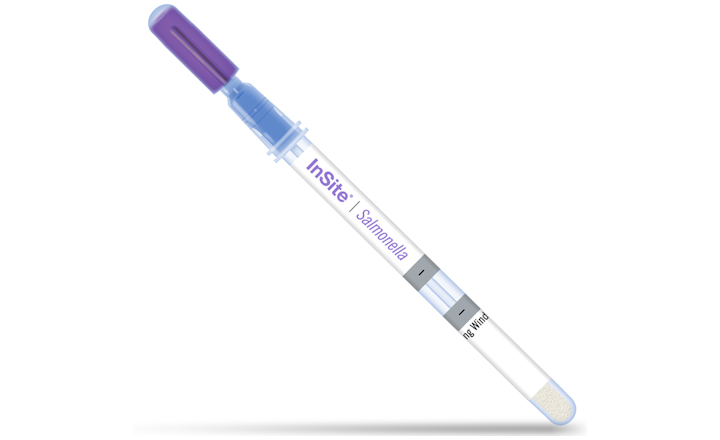
Based on rigorous and independent analyses, the InSite Salmonella device is an easy-to-use test for the screening and detection of Salmonella enterica on food contact surfaces and other processing equipment containing stainless steel, plastic, rubber, ceramic or sealed concrete surfaces after cleaning. It is a self-contained device, which consists of a liquid medium formulated with growth enhancers and chromogenic compounds selective for Salmonella species. All InSite test devices contain neutralizers in the wetting solution to help combat potential sanitizer side effects and improve sample collection.
Product Highlights:
- Simple to swab the environmental test area and incubate
- No expensive lab equipment is required
- Rapid time-to-results of 24-48 hours
- Reduce material and labor
- Self-contained reagent
- Easy to interpret color change (a change in color after incubation is considered presumptive positive for Salmonella enterica)
For more information on Hygiena's InSite Salmonella test device, please visit: InSite Salmonella - Pathogen Detection - Hygiena. Alternatively, use the Request Information button below to make direct contact with Hygiena.