The demand for affordable drugs and increased access to lifesaving medications has driven competition in the biologics market, particularly with the rise of biosimilars. Biosimilars are highly similar to approved biopharmaceutical drugs and can be manufactured once the original product's patent expires. With a growing number of biosimilars entering the market, there is a need for efficient instrumentation and assay formats to expedite development processes.
Unlike small-molecule generics, biosimilars cannot be exact replicas of the reference medicinal product (RMP) due to the inherent variability and complexity of biological medicines. However, biosimilars must demonstrate no clinically meaningful differences in safety, purity, and potency compared to the RMP, with minor variations in inactive components allowed.
The development of biosimilars begins with the creation of multiple cell clones containing the primary amino acid sequence of the desired biologic. Upstream bioprocessing involves optimizing the expression of the desired biologic through cell line and process development. Downstream bioprocessing focuses on isolating, purifying, and characterizing the target biologic from complex mixtures while ensuring purity, potency, and safety standards are met.
Demonstrating biosimilarity throughout upstream and downstream processes can be challenging using traditional analytical methods, leading to the need for alternative high-throughput approaches.
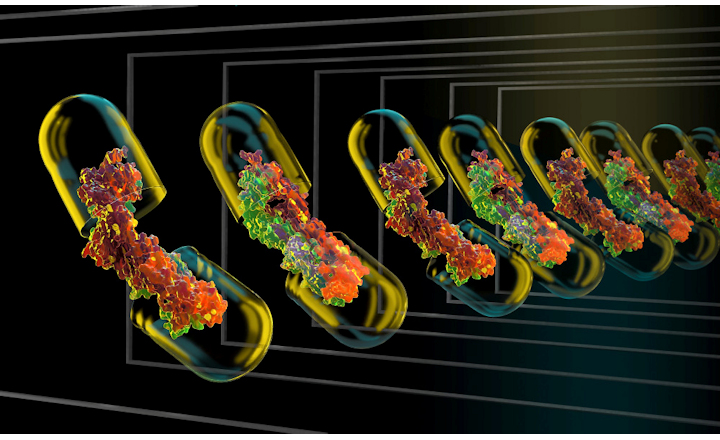
Traditionally, biosimilarity is assessed through comparability studies, where the binding of the proposed biosimilar must fall within a specified range compared to the RMP. Octet® BLI systems offer a high-throughput analytical approach for assessing key biologics attributes, including target binding, Fcγ receptor binding, glycan screening, impurities, and host cell proteins.
This application note focuses on combining a single analyte kinetic screen using an IgG1 antibody against Fcγ receptors CD16a V176 and CD64, as well as the antigen TNFα, to rapidly assess column elution fractions of a biosimilar in development. The approach can be adapted to assess multiple critical quality attributes (CQAs) and requires minimal protein amounts.
By combining kinetic screening with the high-throughput capabilities of Octet® BLI systems, a streamlined approach is achieved. Multiple attributes such as Fcγ receptor binding and glycan screening can be rapidly assessed directly from supernatants, with or without purification, offering a convenient orthogonal approach for biosimilar characterization.
Download the Application Note here