BioTrak® provides real-time counts of total and viable particles in pharmaceutical manufacturing environments to reduce aseptic interventions, with minimal or no human ntervention or the need for additional reagents.
BioTrak® Real-Time Viable Particle Counter integrates an ISO21501-4 compliant particle counter, a state-of-the-art viability detector and a gelatin filter collection medium which can be utilised continuously for up to 9 hours prior to a change, allowing identification of viable organisms capable of growing on Trypticase Soy Agar (TSA), as per current GMP requirements.
BioTrak® is an alternative rapid microbiological method (ARMM) for viable air testing, based on laser induced fluorescence (LIF). The core technology involves detecting viable particles, also called biofluorescent particle counts (BFPC), with sample capture capability and ISO-compliant total particle counting to offer a complete solution for pharmaceutical environmental monitoring.
When combined with Facility Management Software (FMS), data collected meets 21 CFR Part 11 compliance assuring the highest level of data integrity.
Viable Particle Counting with Laser Induced Fluorescence
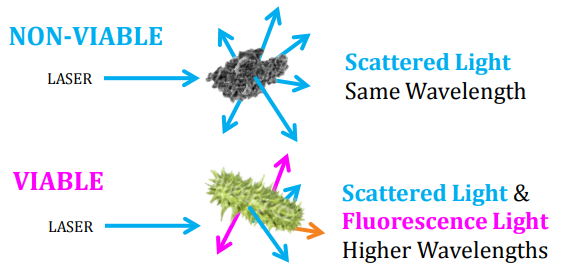
Fluorescence is a good indicator for determining if a particle is viable or non-viable because microbial cells contain relatively high concentrations of molecules that intrinsically fluoresce. As this method relies on optical characteristics rather than on culture-based growth (which depend on media type, incubation temperature, incubation time, etc.), it generally detects more viable particles than by traditional growth-based methods, so it produces a more realistic measurement for viable contamination. BioTrak’s® “fluorescence detection engine” has two channels. This gives a better discrimination amongst different types of auto-fluorescing particles. This minimises false positive detection between interferent particles which are non-biological but may also auto-fluoresce (like paper, IPA, pollen, skin cells, etc) and the microbial particles that we are really interested in. Earlier instruments only had a single fluorescence counting channel and no discrimination amongst auto-fluorescing particles was possible.
Beyond the monitoring of aseptic processes, BioTrak® can be used to provide value as a stand-alone instrument. These applications include routine periodic testing, quickly identifying sources of contamination during Environmental Monitoring investigations, data collection to allow for immediate risk-based room release and for aiding in the identification and mitigation of risk for improved risk assessments.
Alternative Applications for BioTrak®
- Validated In-Process Measurement - long term benefit / goal
- Root Cause Investigations - immediate benefit
- Risk Based Room Release - immediate benefit
- Air Exchange Optimisation - immediate benefit
- Operator Training - immediate benefit
- Comparability Studies - immediate benefit
1. Validated In-Process Measurement
Continuous Automated Monitoring of Aseptic Environments.
Traditional microbial monitoring inside isolators and filling lines can cause disruption to processes and production due to the limited period traditional active air sampling can be used continuously.

Implementing the BioTrak® Real-Time Viable Particle Counter in fill-finish lines eliminates the need for process interruptions for microbial monitoring reasons. Every aseptic fill-finish operation must perform in-process microbial monitoring to comply with Good Manufacturing Practices (GMP). These activities introduce significant risk and negatively impacts throughput. The cost savings achieved using the BioTrak® Real-Time Viable Particle Counter for interruption-free, continuous in-process monitoring can be significant. The cost savings depend on several factors, such as cost of goods manufactured, frequency and duration of microbial monitoring interventions, risk mitigation procedures, and the capacity of the line. During an intervention period, filled drug containers on the line undergo negative exposure, causing them to be rejected as waste. This practice reduces risk but sacrifices production. The actual cost savings achieved with integration of the BioTrak® Real-Time Viable Particle Counter is highly dependent on the products and processes involved.
Biotrak® as part of an Environmental Monitoring System
The BioTrak® Real-Time Viable Particle Counter can be integrated into a facility monitoring system whereby operation, data collection, analysis, alert/alarm functions and batch reporting are fully automated.
The BioTrak® can be integrated into a System that monitors the airborne environment continuously. Every minute a result for total particle counts and viable particle counts will be posted to the validated FMS system.
When pre-configured alert and action limits are exceeded, end users will be immediately notified to enable an immediate response to a viable particle excursion. Viable article counting in real time in conjunction with the FMS software will deliver important information about your processes in real time. With the ability to enable alarms, set off sounders and send e-mail notifications, it gives the customer the ability to react to excursions in a very short time minimising losses due to production waste, storage etc.
2. Root Cause Investigations
When particle deviations occur in Pharmaceutical cleanrooms, root cause investigations are initiated. The extent of any investigation should be consistent with the severity of the excursion. As the FDA Aseptic Processing Guidance 2004 states:
“Routine particle monitoring is useful in rapidly detecting significant deviations in air cleanliness from qualified processing norms (e.g., clean area classification). A result outside the established classification level at a given location should be investigated as to its cause. The extent of investigation should be consistent with the severity of the excursion and include an evaluation of trending data. Appropriate corrective action should be implemented, as necessary, to prevent future deviations.”
The BioTrak® Real-Time Viable Particle Counter can be used to greatly improve the user's ability to identify a definitive root cause in the event of an airborne viable excursion. One factor that plays a large role in this difficult situation is the fact that active air samplers can only offer a snapshot of a very brief period of time in a small area, making detection, a matter of being in the right place at the right time. In contrast, the BioTrak Real-Time Viable Particle Counter has two features that make it an ideal tool to use in root cause investigations:
a. The BioTrak® Real-Time Viable Particle Counter has a scanning feature that can be used to scan equipment, utility penetrations, filters, etc., and provide rapid, real-time indication of a source of viable contamination. This can be set up as an audible signal when microbial activity is detected. This is done in a beep mode setting and microbial organisms beep like a type of “Microbial Geiger Counter”.
b. The BioTrak® Real-Time Viable Particle Counter can be set up to continuously monitor at a
location to capture transient events of elevated viable contamination that can be directly
correlated to the activities being performed in the area.
Scan Suspected Sources
If a possible contamination source is identified by the root cause analysis process, it can be very difficult to confirm the problem using traditional testing methods, in contrast, a filter scanning probe can be used with the BioTrak® Real-Time Viable Particle Counter to scan a source and supply immediate feedback on airborne viable levels from a discrete location.
To perform a scan, a scanning probe needs to be attached to the BioTrak®. A baseline needs to be established using the instrument away from the suspected contaminated area. A 0.5um alarm threshold needs to be set, based on the baseline results.
Scan the suspected area by moving the probe methodically over all areas of interest. Each second, the BioTrak® will divide the actual number of viables detected by the entered threshold and emit a corresponding number of beeps. For example, if the threshold is set at 20 and 60 viable particles are detected; the BioTrak® will beep at a rate of 3 beeps per second. Therefore, the level of viable contamination at a location can be determined by the frequency of the beeps emitted while scanning that location.
Test for Effectivity
Once a root cause has been identified and actions have been taken to correct the issue, testing needs to be performed to confirm that those actions have been effective. Scanning and/or continuous monitoring should be repeated to verify that the issue has been resolved and the area can be immediately returned to use with confidence that the issue has been resolved.
3. Risk Based Room Release
To re-establish a Grade A cleanroom, isolator or R.A.B.S. by traditional monitoring techniques, can take a week or so. However, if the normal at-rest and operational parameters of that space is known prior to the downtime, when BioTrak® indicates that those normal parameters have been reached (measurements being in real time), then the room or cabinet can be released for used, based on risk. This means the space can be released at the earliest convenience.
4. Air Exchange Optimistation
What is the effect of reducing air flow speeds in Grade A environments?
How long does it take for a Grade A space to re-establish itself after the filtration is turned off (for maintenance, or downtime overnight, or at a weekend)?
BioTrak® can help save valuable resources and expense by indicating how long a space takes to re-establish and what the effect of running at a lesser air exchange rate. Optimum rates can be established.
5. Operator Training
Aseptic gowning procedures for operatives working in a GMP Grade A aseptic manufacturing suite, need to be successfully demonstrated and approved as part of a training regime and documented in a Training Record before that operative can be approved for entrance into the Grade A work area.
Some clients employ a body booth enclosure with a BioTrak® attached to the air outlet and as an operative walks through the enclosure and demonstrate that numbers of viable particles detected comply with accepted limits. Acceptable limits can be inputted into BioTrak’s® action / alert alarms to demonstrate successful gowning, or aid with retraining if sterile techniques need improving.
6. Comparability Studies
When a Rapid Method is being considered, GMP (Annex 1) Guidelines state that the Rapid Method needs to be demonstrated to be at least as effective as the traditional method currently being employed. Comparison studies with the BioTrak® and active air samplers / optical particle counters can be set out in an approved Standard Operating Procedure (SOP) outlining the test procedure and results can be compared prior to any rapid method being put into operation.
The flow rate of the air sampler and particle counter should be compared to BioTrak’s® (28.3 LPM) and normalised so sample sizes are equivalent. Documents are available to perform this type of Comparability Study and help is on hand by trained BioTrak® Application Engineers
Conclusion
Traditional microbial active air sampling and compendial culture-based count methods have been used for decades, providing invaluable microbial contamination information. However, they typically require a delay of up to 5 days for incubation and analysis — which could result in production of potentially contaminated product and expensive batch wastage.
The BioTrak® Real-Time Viable Particle Counter, provides information on when contamination excursions occur, as they happen. Meaning you can respond to an event right away by segregating potentially contaminated products and immediately locating the source of potential contamination by using the Biotrak for root cause analysis. This minimizes product loss and enhances product safety.
For more information use the 'request information' button below.
Reference: TSI Inc various papers available on request

























