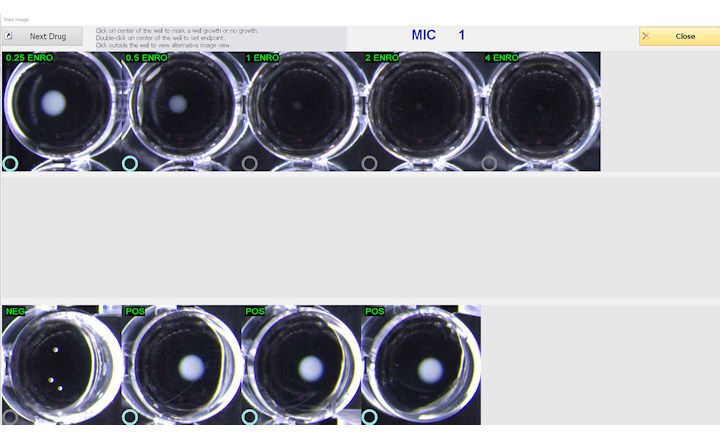
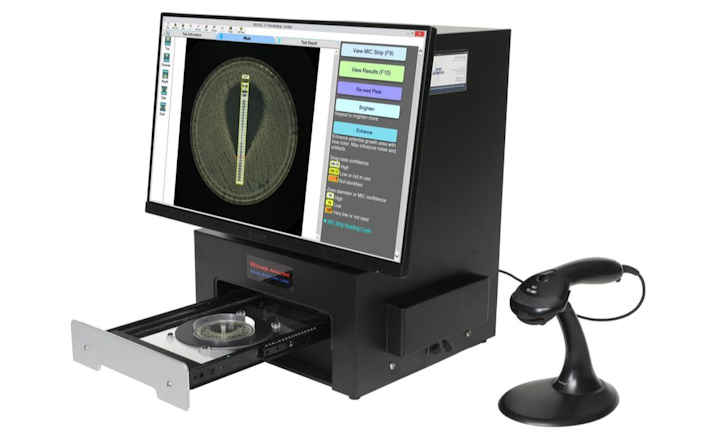
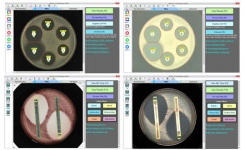
The BIOMIC V3 Microbiology System digitally reads, interprets, and records standard AMR tests including antibiotic disk diffusion, 96-well microtiter, and Etest/MIC Strips. BIOMIC V3 is currently utilized in AMR surveillance programs including CDC Carbapenem Resistance (CRE), CDC Gonococcal Isolate Surveillance Program, and US-FDA Vet-LIRN Veterinary Laboratory Investigation and Response Network.
AMR advantages with BIOMIC V3 include custom data export, saved test images, no routine system maintenance, current CLSI and EUCAST guidelines, custom reports, online training, verified QC testing, and standardized test procedures.
Giles Scientific has extensive experience in global AMR programs. From 1997-2010, 151 hospital labs in 40 countries with BIOMIC V3 systems participated in the ARTEMIS drug resistance study sponsored by Pfizer. 50+ journal publications demonstrate BIOMIC V3's testing and AMR surveillance capabilities.
Visit www.biomic.com