| Alicyclobacillus are sometimes called acidophilic thermophilic bacteria (ATB). As the name implies, ATB grow well in acidic environments, surviving at pH levels as low as 2.5. The bacteria also grow well at elevated temperatures. The spores can survive the pasteurization treatment given to most shelf stable, glass packaged fruit juices. The heat treatment activates the spores to begin growth. Most contamination problems show up s everal days after bottling. The organisms tend to be resistant to Sorbate and SO2.
Research has shown that the off-odor defect in pasteurized apple juice is due to guaiacol, produced by ATB contamination from vanillin in the juice. The odor of guaiacol is quite distinctive and a clear sign that there is a spoilage problem due to ATB. Guaiacol detection in finished juice products is accomplished by methylene chloride extraction, concentration and analysis by GC-MS.
Studies show that Alicyclobacillus grows well in apple juice, tomato juice, white grape juice and various blends of juices (Splittstoesser, et al., 1994; Splittstoesser et al., 1998). The bacteria have been isolated from citrus processing lines and are potential spoilage agents of beverages containing citrus juices (Wisse and Parish, 1998).
Some studies have used Potato Dextrose Agar (PDA), pH 3.5 as a selective medium for Alicyclobacillus. The juice or other sample is heated at 60&#;176;C for 60 minutes prior to culturing to destroy non-heat resistant organisms such as yeasts and to activate dormant spores. The plates are incubated 5 to 7 days at 43°C. Media used by other investigators include K Media (Walls and Chuyate, 1998) and ALI Agar (Wisse and Parish, 1998). Because only low populations of the spores may be present, detection often requires culturing large volumes of juice. When the juice is filterable, several hundred milliliters may be passed through 0.45 um membrane filters. ATB can be detected in spoiled products by the pour plate method or membrane filtration method, with growth on K Agar medium. Concentrates are analyzed for Alicyclobacillus spores by treating the sample to a preliminary heat shock, followed by plating on K Agar medium. Heat resistance studies have shown that Alicyclobacillus spores can survive the usual hot fill processes that are given to commercial juices. The data also shows that increasing the Brix makes the spores more resistant to heat. The spores are less resistant as the pH is decreased and the type of organic acid can be important (Pontius et al., 1998).
Using the BioSys Instrument to Detect Alicyclobacillus
The ViV (vial-in-vial) system consists of two containers, an internal vial and an external vial. Organisms grow in the liquid medium in the internal vial and generate carbon-dioxide gas from their metabolic activity. Since the internal vial is placed unsealed into the external vial containing dye indicator, the generated gas is released from the internal vial to fill the headspace of the external vial. The generated gas travels to the surface of the indicator and interacts with its chemical compounds. The generated carbon dioxide interacts with the KOH compound to lower its pH. Thymolphthalein dye changes from purple at pH=9 (or higher) to colorless at lower pH values. The BioSys instrument records the time taken for an identified change in the absorban ce. This will occur when the CO2 that is released by growing organisms is absorbed by the KOH in the external vial, resulting in a pH change and a concurrent change in dye color.
Method
Several strains of Alicyclobacillus were tested in modified K Broth developed for the BioSys system. For each strain, 0.1ml of a suspension containing 100-1,000 cfu/ml was introduced into the modified K Broth in the internal vial. The internal vials were inserted into the external vials containing dye indicator. The cap was sealed tightly and the vial inserted into the BioSys instrument. The vials were monitored for 36 hours.
Algorithm Parameters
The following temperature and algorithm parameters were used in the detection of Alicyclobacillus:
| Vial | Threshold | Skip | >Shuteye | Duration | Temperature | | ViV | 10 | 1 | 25 | 36 | 43°C | Curves Obtained
A typical curve and clear detection time in the BioSys instrument was observed for the presence of Alicyclobacillus in all cases. Typical curves obtained are shown below:
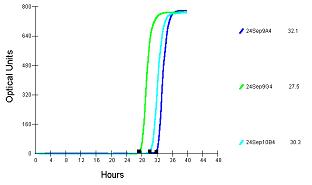 Key: Culture Detection Time (hrs) cfu/ml
Alicyclobacillus sp. 231-471-1 32.1 5.8 x 101
Alicyclobacillus sp. 231-47 6-6 27.5 2.74 x 102
Alicyclobacillus sp. K-AJ 30.3 3.4 x 101
Positive and Negative Vials
The figure at the top of the page, shows a vial with Alicyclobacillus in the left and a vial with a sample that did not contain the organisms on the right.
Conclusion:
After heat shock, product samples can be inoculated or membrane filters inserted into the BioSys ViV inner vial containing modified K Broth Medium. The inner vial is inserted into the outer vial, which is tightly capped, placed in the BioSys instrument and monitored for 36 hours in the system. If the sample contains the target organism, the computerized system will detect the presence of the organism and will alert the user in "real time" of such occurrence. Samples that do not detect within 36 hours are negative and do not contain Alicyclobacillus.
References Cerny, G., W. Hennlich, K. Poral la. 1984. Spoilage of fruit juice by bacilli: isolation and characterization of the spoilage organism. Z. Lebensm. Unters. Forsch. 179:224-227.
Pontius, A. J., J. E. Rushing, and P. M. Foegeding. 1998. Heat resistance of Alicyclobacillus acidoterrestris spores as affected by various pH values and organic acids. J. Food Protection 61:41-46.
Splittstoesser, D. F. 1993. Recent developments in the microbiology of fruit juices, p. 59-61. In D. L. Downing (ed), Juice Technology Workshop, Report 67, NYS Agric. Expt. Station, Geneva, NY
Splittstoesser, D. F., J. J. Churey, and C. Y. Lee. 1994. Growth characteristics of aciduric sporeforming bacteria isolated from fruit juices. J. Food Protection 57:1080.
Splittstoesser, D. F., and J. J. Churey. 1996. Unique spoilage organisms of musts and wines, p 36-41. In T. Toland and K. C. Fugelsang (ed), Wine spoilage microbiology Conference. California State University, Fresno.
Splittstoesser, D. F., C. Y. Lee and J. J. Churey. 1998. Control of Alicyclobacillus in the juice industry. Dairy Food and Environ. Sanitation 18:585-587.
Walls, I. And R. Chuyate, 1998. Alicyclobacillus - Historical perspective and preliminary characterization study. Dairy Food and Environ. Sanitation 18:499-503.
Wisotzkey, J. D., P. Jurtshuk, Jr., G. E. Fox, G. Deinhard, and K. Poralla. 1992. Comparative sequence analysis on the 16S (rDNA) of Bacillus acidocaldarius, Bacillus acidoterrestris and Bacillus cycloheptanicus and a proposal for creation of a new genus Alicyclobacillus gen. nov. Intl. J. Systematic Bacteriol. 42:263-269
Wisse, C. A., and M. E. Parish. 1998. Isolation and enumeration of sporeforming, thermoacidophilic, rod-shaped bacteria from citrus processing environments. . Dairy Food and Environ. Sanitation 18:504-509.
|

















